- Research article
- Open access
- Published: 04 June 2021

Coronavirus disease (COVID-19) pandemic: an overview of systematic reviews
- Israel Júnior Borges do Nascimento 1 , 2 ,
- Dónal P. O’Mathúna 3 , 4 ,
- Thilo Caspar von Groote 5 ,
- Hebatullah Mohamed Abdulazeem 6 ,
- Ishanka Weerasekara 7 , 8 ,
- Ana Marusic 9 ,
- Livia Puljak ORCID: orcid.org/0000-0002-8467-6061 10 ,
- Vinicius Tassoni Civile 11 ,
- Irena Zakarija-Grkovic 9 ,
- Tina Poklepovic Pericic 9 ,
- Alvaro Nagib Atallah 11 ,
- Santino Filoso 12 ,
- Nicola Luigi Bragazzi 13 &
- Milena Soriano Marcolino 1
On behalf of the International Network of Coronavirus Disease 2019 (InterNetCOVID-19)
BMC Infectious Diseases volume 21 , Article number: 525 ( 2021 ) Cite this article
16k Accesses
28 Citations
13 Altmetric
Metrics details
Navigating the rapidly growing body of scientific literature on the SARS-CoV-2 pandemic is challenging, and ongoing critical appraisal of this output is essential. We aimed to summarize and critically appraise systematic reviews of coronavirus disease (COVID-19) in humans that were available at the beginning of the pandemic.
Nine databases (Medline, EMBASE, Cochrane Library, CINAHL, Web of Sciences, PDQ-Evidence, WHO’s Global Research, LILACS, and Epistemonikos) were searched from December 1, 2019, to March 24, 2020. Systematic reviews analyzing primary studies of COVID-19 were included. Two authors independently undertook screening, selection, extraction (data on clinical symptoms, prevalence, pharmacological and non-pharmacological interventions, diagnostic test assessment, laboratory, and radiological findings), and quality assessment (AMSTAR 2). A meta-analysis was performed of the prevalence of clinical outcomes.
Eighteen systematic reviews were included; one was empty (did not identify any relevant study). Using AMSTAR 2, confidence in the results of all 18 reviews was rated as “critically low”. Identified symptoms of COVID-19 were (range values of point estimates): fever (82–95%), cough with or without sputum (58–72%), dyspnea (26–59%), myalgia or muscle fatigue (29–51%), sore throat (10–13%), headache (8–12%) and gastrointestinal complaints (5–9%). Severe symptoms were more common in men. Elevated C-reactive protein and lactate dehydrogenase, and slightly elevated aspartate and alanine aminotransferase, were commonly described. Thrombocytopenia and elevated levels of procalcitonin and cardiac troponin I were associated with severe disease. A frequent finding on chest imaging was uni- or bilateral multilobar ground-glass opacity. A single review investigated the impact of medication (chloroquine) but found no verifiable clinical data. All-cause mortality ranged from 0.3 to 13.9%.
Conclusions
In this overview of systematic reviews, we analyzed evidence from the first 18 systematic reviews that were published after the emergence of COVID-19. However, confidence in the results of all reviews was “critically low”. Thus, systematic reviews that were published early on in the pandemic were of questionable usefulness. Even during public health emergencies, studies and systematic reviews should adhere to established methodological standards.
Peer Review reports
The spread of the “Severe Acute Respiratory Coronavirus 2” (SARS-CoV-2), the causal agent of COVID-19, was characterized as a pandemic by the World Health Organization (WHO) in March 2020 and has triggered an international public health emergency [ 1 ]. The numbers of confirmed cases and deaths due to COVID-19 are rapidly escalating, counting in millions [ 2 ], causing massive economic strain, and escalating healthcare and public health expenses [ 3 , 4 ].
The research community has responded by publishing an impressive number of scientific reports related to COVID-19. The world was alerted to the new disease at the beginning of 2020 [ 1 ], and by mid-March 2020, more than 2000 articles had been published on COVID-19 in scholarly journals, with 25% of them containing original data [ 5 ]. The living map of COVID-19 evidence, curated by the Evidence for Policy and Practice Information and Co-ordinating Centre (EPPI-Centre), contained more than 40,000 records by February 2021 [ 6 ]. More than 100,000 records on PubMed were labeled as “SARS-CoV-2 literature, sequence, and clinical content” by February 2021 [ 7 ].
Due to publication speed, the research community has voiced concerns regarding the quality and reproducibility of evidence produced during the COVID-19 pandemic, warning of the potential damaging approach of “publish first, retract later” [ 8 ]. It appears that these concerns are not unfounded, as it has been reported that COVID-19 articles were overrepresented in the pool of retracted articles in 2020 [ 9 ]. These concerns about inadequate evidence are of major importance because they can lead to poor clinical practice and inappropriate policies [ 10 ].
Systematic reviews are a cornerstone of today’s evidence-informed decision-making. By synthesizing all relevant evidence regarding a particular topic, systematic reviews reflect the current scientific knowledge. Systematic reviews are considered to be at the highest level in the hierarchy of evidence and should be used to make informed decisions. However, with high numbers of systematic reviews of different scope and methodological quality being published, overviews of multiple systematic reviews that assess their methodological quality are essential [ 11 , 12 , 13 ]. An overview of systematic reviews helps identify and organize the literature and highlights areas of priority in decision-making.
In this overview of systematic reviews, we aimed to summarize and critically appraise systematic reviews of coronavirus disease (COVID-19) in humans that were available at the beginning of the pandemic.
Methodology
Research question.
This overview’s primary objective was to summarize and critically appraise systematic reviews that assessed any type of primary clinical data from patients infected with SARS-CoV-2. Our research question was purposefully broad because we wanted to analyze as many systematic reviews as possible that were available early following the COVID-19 outbreak.
Study design
We conducted an overview of systematic reviews. The idea for this overview originated in a protocol for a systematic review submitted to PROSPERO (CRD42020170623), which indicated a plan to conduct an overview.
Overviews of systematic reviews use explicit and systematic methods for searching and identifying multiple systematic reviews addressing related research questions in the same field to extract and analyze evidence across important outcomes. Overviews of systematic reviews are in principle similar to systematic reviews of interventions, but the unit of analysis is a systematic review [ 14 , 15 , 16 ].
We used the overview methodology instead of other evidence synthesis methods to allow us to collate and appraise multiple systematic reviews on this topic, and to extract and analyze their results across relevant topics [ 17 ]. The overview and meta-analysis of systematic reviews allowed us to investigate the methodological quality of included studies, summarize results, and identify specific areas of available or limited evidence, thereby strengthening the current understanding of this novel disease and guiding future research [ 13 ].
A reporting guideline for overviews of reviews is currently under development, i.e., Preferred Reporting Items for Overviews of Reviews (PRIOR) [ 18 ]. As the PRIOR checklist is still not published, this study was reported following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2009 statement [ 19 ]. The methodology used in this review was adapted from the Cochrane Handbook for Systematic Reviews of Interventions and also followed established methodological considerations for analyzing existing systematic reviews [ 14 ].
Approval of a research ethics committee was not necessary as the study analyzed only publicly available articles.
Eligibility criteria
Systematic reviews were included if they analyzed primary data from patients infected with SARS-CoV-2 as confirmed by RT-PCR or another pre-specified diagnostic technique. Eligible reviews covered all topics related to COVID-19 including, but not limited to, those that reported clinical symptoms, diagnostic methods, therapeutic interventions, laboratory findings, or radiological results. Both full manuscripts and abbreviated versions, such as letters, were eligible.
No restrictions were imposed on the design of the primary studies included within the systematic reviews, the last search date, whether the review included meta-analyses or language. Reviews related to SARS-CoV-2 and other coronaviruses were eligible, but from those reviews, we analyzed only data related to SARS-CoV-2.
No consensus definition exists for a systematic review [ 20 ], and debates continue about the defining characteristics of a systematic review [ 21 ]. Cochrane’s guidance for overviews of reviews recommends setting pre-established criteria for making decisions around inclusion [ 14 ]. That is supported by a recent scoping review about guidance for overviews of systematic reviews [ 22 ].
Thus, for this study, we defined a systematic review as a research report which searched for primary research studies on a specific topic using an explicit search strategy, had a detailed description of the methods with explicit inclusion criteria provided, and provided a summary of the included studies either in narrative or quantitative format (such as a meta-analysis). Cochrane and non-Cochrane systematic reviews were considered eligible for inclusion, with or without meta-analysis, and regardless of the study design, language restriction and methodology of the included primary studies. To be eligible for inclusion, reviews had to be clearly analyzing data related to SARS-CoV-2 (associated or not with other viruses). We excluded narrative reviews without those characteristics as these are less likely to be replicable and are more prone to bias.
Scoping reviews and rapid reviews were eligible for inclusion in this overview if they met our pre-defined inclusion criteria noted above. We included reviews that addressed SARS-CoV-2 and other coronaviruses if they reported separate data regarding SARS-CoV-2.
Information sources
Nine databases were searched for eligible records published between December 1, 2019, and March 24, 2020: Cochrane Database of Systematic Reviews via Cochrane Library, PubMed, EMBASE, CINAHL (Cumulative Index to Nursing and Allied Health Literature), Web of Sciences, LILACS (Latin American and Caribbean Health Sciences Literature), PDQ-Evidence, WHO’s Global Research on Coronavirus Disease (COVID-19), and Epistemonikos.
The comprehensive search strategy for each database is provided in Additional file 1 and was designed and conducted in collaboration with an information specialist. All retrieved records were primarily processed in EndNote, where duplicates were removed, and records were then imported into the Covidence platform [ 23 ]. In addition to database searches, we screened reference lists of reviews included after screening records retrieved via databases.
Study selection
All searches, screening of titles and abstracts, and record selection, were performed independently by two investigators using the Covidence platform [ 23 ]. Articles deemed potentially eligible were retrieved for full-text screening carried out independently by two investigators. Discrepancies at all stages were resolved by consensus. During the screening, records published in languages other than English were translated by a native/fluent speaker.
Data collection process
We custom designed a data extraction table for this study, which was piloted by two authors independently. Data extraction was performed independently by two authors. Conflicts were resolved by consensus or by consulting a third researcher.
We extracted the following data: article identification data (authors’ name and journal of publication), search period, number of databases searched, population or settings considered, main results and outcomes observed, and number of participants. From Web of Science (Clarivate Analytics, Philadelphia, PA, USA), we extracted journal rank (quartile) and Journal Impact Factor (JIF).
We categorized the following as primary outcomes: all-cause mortality, need for and length of mechanical ventilation, length of hospitalization (in days), admission to intensive care unit (yes/no), and length of stay in the intensive care unit.
The following outcomes were categorized as exploratory: diagnostic methods used for detection of the virus, male to female ratio, clinical symptoms, pharmacological and non-pharmacological interventions, laboratory findings (full blood count, liver enzymes, C-reactive protein, d-dimer, albumin, lipid profile, serum electrolytes, blood vitamin levels, glucose levels, and any other important biomarkers), and radiological findings (using radiography, computed tomography, magnetic resonance imaging or ultrasound).
We also collected data on reporting guidelines and requirements for the publication of systematic reviews and meta-analyses from journal websites where included reviews were published.
Quality assessment in individual reviews
Two researchers independently assessed the reviews’ quality using the “A MeaSurement Tool to Assess Systematic Reviews 2 (AMSTAR 2)”. We acknowledge that the AMSTAR 2 was created as “a critical appraisal tool for systematic reviews that include randomized or non-randomized studies of healthcare interventions, or both” [ 24 ]. However, since AMSTAR 2 was designed for systematic reviews of intervention trials, and we included additional types of systematic reviews, we adjusted some AMSTAR 2 ratings and reported these in Additional file 2 .
Adherence to each item was rated as follows: yes, partial yes, no, or not applicable (such as when a meta-analysis was not conducted). The overall confidence in the results of the review is rated as “critically low”, “low”, “moderate” or “high”, according to the AMSTAR 2 guidance based on seven critical domains, which are items 2, 4, 7, 9, 11, 13, 15 as defined by AMSTAR 2 authors [ 24 ]. We reported our adherence ratings for transparency of our decision with accompanying explanations, for each item, in each included review.
One of the included systematic reviews was conducted by some members of this author team [ 25 ]. This review was initially assessed independently by two authors who were not co-authors of that review to prevent the risk of bias in assessing this study.
Synthesis of results
For data synthesis, we prepared a table summarizing each systematic review. Graphs illustrating the mortality rate and clinical symptoms were created. We then prepared a narrative summary of the methods, findings, study strengths, and limitations.
For analysis of the prevalence of clinical outcomes, we extracted data on the number of events and the total number of patients to perform proportional meta-analysis using RStudio© software, with the “meta” package (version 4.9–6), using the “metaprop” function for reviews that did not perform a meta-analysis, excluding case studies because of the absence of variance. For reviews that did not perform a meta-analysis, we presented pooled results of proportions with their respective confidence intervals (95%) by the inverse variance method with a random-effects model, using the DerSimonian-Laird estimator for τ 2 . We adjusted data using Freeman-Tukey double arcosen transformation. Confidence intervals were calculated using the Clopper-Pearson method for individual studies. We created forest plots using the RStudio© software, with the “metafor” package (version 2.1–0) and “forest” function.
Managing overlapping systematic reviews
Some of the included systematic reviews that address the same or similar research questions may include the same primary studies in overviews. Including such overlapping reviews may introduce bias when outcome data from the same primary study are included in the analyses of an overview multiple times. Thus, in summaries of evidence, multiple-counting of the same outcome data will give data from some primary studies too much influence [ 14 ]. In this overview, we did not exclude overlapping systematic reviews because, according to Cochrane’s guidance, it may be appropriate to include all relevant reviews’ results if the purpose of the overview is to present and describe the current body of evidence on a topic [ 14 ]. To avoid any bias in summary estimates associated with overlapping reviews, we generated forest plots showing data from individual systematic reviews, but the results were not pooled because some primary studies were included in multiple reviews.
Our search retrieved 1063 publications, of which 175 were duplicates. Most publications were excluded after the title and abstract analysis ( n = 860). Among the 28 studies selected for full-text screening, 10 were excluded for the reasons described in Additional file 3 , and 18 were included in the final analysis (Fig. 1 ) [ 25 , 26 , 27 , 28 , 29 , 30 , 31 , 32 , 33 , 34 , 35 , 36 , 37 , 38 , 39 , 40 , 41 , 42 ]. Reference list screening did not retrieve any additional systematic reviews.
PRISMA flow diagram
Characteristics of included reviews
Summary features of 18 systematic reviews are presented in Table 1 . They were published in 14 different journals. Only four of these journals had specific requirements for systematic reviews (with or without meta-analysis): European Journal of Internal Medicine, Journal of Clinical Medicine, Ultrasound in Obstetrics and Gynecology, and Clinical Research in Cardiology . Two journals reported that they published only invited reviews ( Journal of Medical Virology and Clinica Chimica Acta ). Three systematic reviews in our study were published as letters; one was labeled as a scoping review and another as a rapid review (Table 2 ).
All reviews were published in English, in first quartile (Q1) journals, with JIF ranging from 1.692 to 6.062. One review was empty, meaning that its search did not identify any relevant studies; i.e., no primary studies were included [ 36 ]. The remaining 17 reviews included 269 unique studies; the majority ( N = 211; 78%) were included in only a single review included in our study (range: 1 to 12). Primary studies included in the reviews were published between December 2019 and March 18, 2020, and comprised case reports, case series, cohorts, and other observational studies. We found only one review that included randomized clinical trials [ 38 ]. In the included reviews, systematic literature searches were performed from 2019 (entire year) up to March 9, 2020. Ten systematic reviews included meta-analyses. The list of primary studies found in the included systematic reviews is shown in Additional file 4 , as well as the number of reviews in which each primary study was included.
Population and study designs
Most of the reviews analyzed data from patients with COVID-19 who developed pneumonia, acute respiratory distress syndrome (ARDS), or any other correlated complication. One review aimed to evaluate the effectiveness of using surgical masks on preventing transmission of the virus [ 36 ], one review was focused on pediatric patients [ 34 ], and one review investigated COVID-19 in pregnant women [ 37 ]. Most reviews assessed clinical symptoms, laboratory findings, or radiological results.
Systematic review findings
The summary of findings from individual reviews is shown in Table 2 . Overall, all-cause mortality ranged from 0.3 to 13.9% (Fig. 2 ).
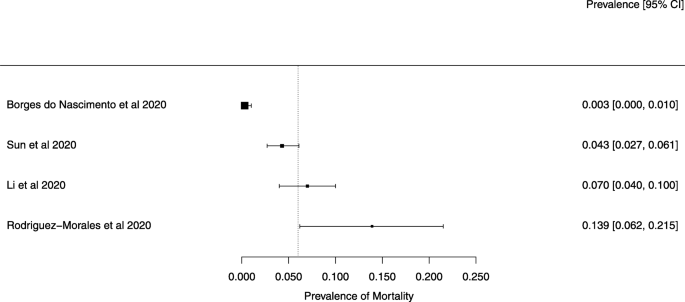
A meta-analysis of the prevalence of mortality
Clinical symptoms
Seven reviews described the main clinical manifestations of COVID-19 [ 26 , 28 , 29 , 34 , 35 , 39 , 41 ]. Three of them provided only a narrative discussion of symptoms [ 26 , 34 , 35 ]. In the reviews that performed a statistical analysis of the incidence of different clinical symptoms, symptoms in patients with COVID-19 were (range values of point estimates): fever (82–95%), cough with or without sputum (58–72%), dyspnea (26–59%), myalgia or muscle fatigue (29–51%), sore throat (10–13%), headache (8–12%), gastrointestinal disorders, such as diarrhea, nausea or vomiting (5.0–9.0%), and others (including, in one study only: dizziness 12.1%) (Figs. 3 , 4 , 5 , 6 , 7 , 8 and 9 ). Three reviews assessed cough with and without sputum together; only one review assessed sputum production itself (28.5%).
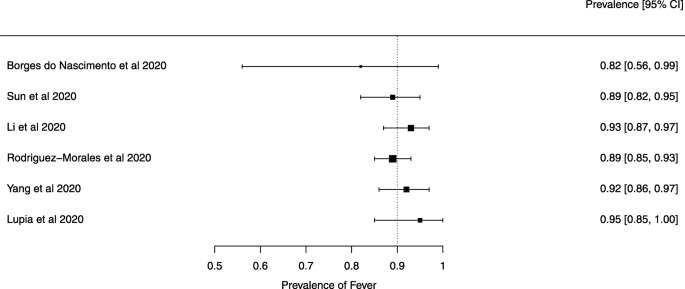
A meta-analysis of the prevalence of fever
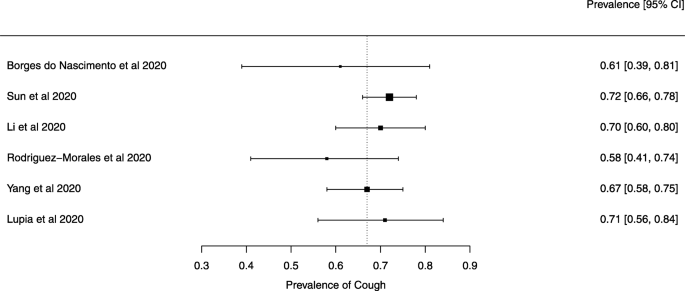
A meta-analysis of the prevalence of cough
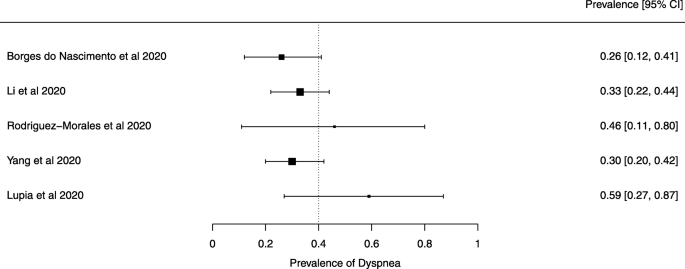
A meta-analysis of the prevalence of dyspnea
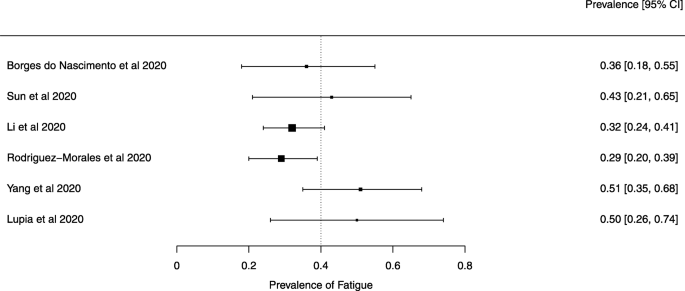
A meta-analysis of the prevalence of fatigue or myalgia
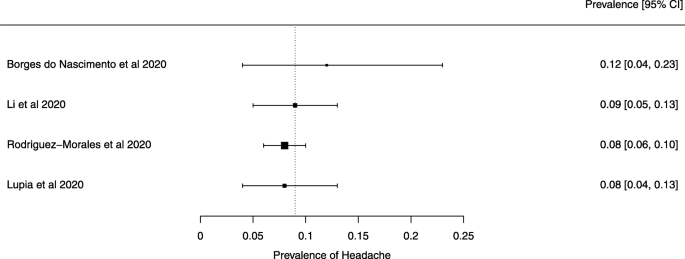
A meta-analysis of the prevalence of headache
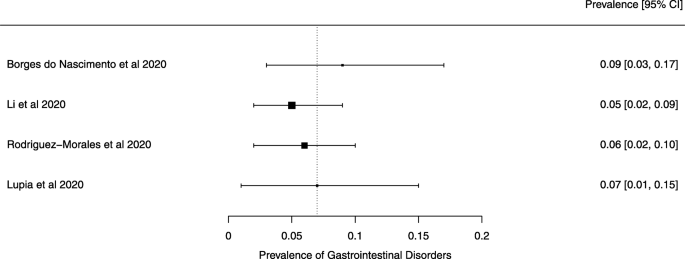
A meta-analysis of the prevalence of gastrointestinal disorders
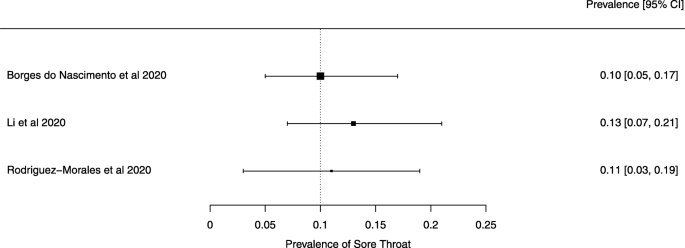
A meta-analysis of the prevalence of sore throat
Diagnostic aspects
Three reviews described methodologies, protocols, and tools used for establishing the diagnosis of COVID-19 [ 26 , 34 , 38 ]. The use of respiratory swabs (nasal or pharyngeal) or blood specimens to assess the presence of SARS-CoV-2 nucleic acid using RT-PCR assays was the most commonly used diagnostic method mentioned in the included studies. These diagnostic tests have been widely used, but their precise sensitivity and specificity remain unknown. One review included a Chinese study with clinical diagnosis with no confirmation of SARS-CoV-2 infection (patients were diagnosed with COVID-19 if they presented with at least two symptoms suggestive of COVID-19, together with laboratory and chest radiography abnormalities) [ 34 ].
Therapeutic possibilities
Pharmacological and non-pharmacological interventions (supportive therapies) used in treating patients with COVID-19 were reported in five reviews [ 25 , 27 , 34 , 35 , 38 ]. Antivirals used empirically for COVID-19 treatment were reported in seven reviews [ 25 , 27 , 34 , 35 , 37 , 38 , 41 ]; most commonly used were protease inhibitors (lopinavir, ritonavir, darunavir), nucleoside reverse transcriptase inhibitor (tenofovir), nucleotide analogs (remdesivir, galidesivir, ganciclovir), and neuraminidase inhibitors (oseltamivir). Umifenovir, a membrane fusion inhibitor, was investigated in two studies [ 25 , 35 ]. Possible supportive interventions analyzed were different types of oxygen supplementation and breathing support (invasive or non-invasive ventilation) [ 25 ]. The use of antibiotics, both empirically and to treat secondary pneumonia, was reported in six studies [ 25 , 26 , 27 , 34 , 35 , 38 ]. One review specifically assessed evidence on the efficacy and safety of the anti-malaria drug chloroquine [ 27 ]. It identified 23 ongoing trials investigating the potential of chloroquine as a therapeutic option for COVID-19, but no verifiable clinical outcomes data. The use of mesenchymal stem cells, antifungals, and glucocorticoids were described in four reviews [ 25 , 34 , 35 , 38 ].
Laboratory and radiological findings
Of the 18 reviews included in this overview, eight analyzed laboratory parameters in patients with COVID-19 [ 25 , 29 , 30 , 32 , 33 , 34 , 35 , 39 ]; elevated C-reactive protein levels, associated with lymphocytopenia, elevated lactate dehydrogenase, as well as slightly elevated aspartate and alanine aminotransferase (AST, ALT) were commonly described in those eight reviews. Lippi et al. assessed cardiac troponin I (cTnI) [ 25 ], procalcitonin [ 32 ], and platelet count [ 33 ] in COVID-19 patients. Elevated levels of procalcitonin [ 32 ] and cTnI [ 30 ] were more likely to be associated with a severe disease course (requiring intensive care unit admission and intubation). Furthermore, thrombocytopenia was frequently observed in patients with complicated COVID-19 infections [ 33 ].
Chest imaging (chest radiography and/or computed tomography) features were assessed in six reviews, all of which described a frequent pattern of local or bilateral multilobar ground-glass opacity [ 25 , 34 , 35 , 39 , 40 , 41 ]. Those six reviews showed that septal thickening, bronchiectasis, pleural and cardiac effusions, halo signs, and pneumothorax were observed in patients suffering from COVID-19.
Quality of evidence in individual systematic reviews
Table 3 shows the detailed results of the quality assessment of 18 systematic reviews, including the assessment of individual items and summary assessment. A detailed explanation for each decision in each review is available in Additional file 5 .
Using AMSTAR 2 criteria, confidence in the results of all 18 reviews was rated as “critically low” (Table 3 ). Common methodological drawbacks were: omission of prospective protocol submission or publication; use of inappropriate search strategy: lack of independent and dual literature screening and data-extraction (or methodology unclear); absence of an explanation for heterogeneity among the studies included; lack of reasons for study exclusion (or rationale unclear).
Risk of bias assessment, based on a reported methodological tool, and quality of evidence appraisal, in line with the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) method, were reported only in one review [ 25 ]. Five reviews presented a table summarizing bias, using various risk of bias tools [ 25 , 29 , 39 , 40 , 41 ]. One review analyzed “study quality” [ 37 ]. One review mentioned the risk of bias assessment in the methodology but did not provide any related analysis [ 28 ].
This overview of systematic reviews analyzed the first 18 systematic reviews published after the onset of the COVID-19 pandemic, up to March 24, 2020, with primary studies involving more than 60,000 patients. Using AMSTAR-2, we judged that our confidence in all those reviews was “critically low”. Ten reviews included meta-analyses. The reviews presented data on clinical manifestations, laboratory and radiological findings, and interventions. We found no systematic reviews on the utility of diagnostic tests.
Symptoms were reported in seven reviews; most of the patients had a fever, cough, dyspnea, myalgia or muscle fatigue, and gastrointestinal disorders such as diarrhea, nausea, or vomiting. Olfactory dysfunction (anosmia or dysosmia) has been described in patients infected with COVID-19 [ 43 ]; however, this was not reported in any of the reviews included in this overview. During the SARS outbreak in 2002, there were reports of impairment of the sense of smell associated with the disease [ 44 , 45 ].
The reported mortality rates ranged from 0.3 to 14% in the included reviews. Mortality estimates are influenced by the transmissibility rate (basic reproduction number), availability of diagnostic tools, notification policies, asymptomatic presentations of the disease, resources for disease prevention and control, and treatment facilities; variability in the mortality rate fits the pattern of emerging infectious diseases [ 46 ]. Furthermore, the reported cases did not consider asymptomatic cases, mild cases where individuals have not sought medical treatment, and the fact that many countries had limited access to diagnostic tests or have implemented testing policies later than the others. Considering the lack of reviews assessing diagnostic testing (sensitivity, specificity, and predictive values of RT-PCT or immunoglobulin tests), and the preponderance of studies that assessed only symptomatic individuals, considerable imprecision around the calculated mortality rates existed in the early stage of the COVID-19 pandemic.
Few reviews included treatment data. Those reviews described studies considered to be at a very low level of evidence: usually small, retrospective studies with very heterogeneous populations. Seven reviews analyzed laboratory parameters; those reviews could have been useful for clinicians who attend patients suspected of COVID-19 in emergency services worldwide, such as assessing which patients need to be reassessed more frequently.
All systematic reviews scored poorly on the AMSTAR 2 critical appraisal tool for systematic reviews. Most of the original studies included in the reviews were case series and case reports, impacting the quality of evidence. Such evidence has major implications for clinical practice and the use of these reviews in evidence-based practice and policy. Clinicians, patients, and policymakers can only have the highest confidence in systematic review findings if high-quality systematic review methodologies are employed. The urgent need for information during a pandemic does not justify poor quality reporting.
We acknowledge that there are numerous challenges associated with analyzing COVID-19 data during a pandemic [ 47 ]. High-quality evidence syntheses are needed for decision-making, but each type of evidence syntheses is associated with its inherent challenges.
The creation of classic systematic reviews requires considerable time and effort; with massive research output, they quickly become outdated, and preparing updated versions also requires considerable time. A recent study showed that updates of non-Cochrane systematic reviews are published a median of 5 years after the publication of the previous version [ 48 ].
Authors may register a review and then abandon it [ 49 ], but the existence of a public record that is not updated may lead other authors to believe that the review is still ongoing. A quarter of Cochrane review protocols remains unpublished as completed systematic reviews 8 years after protocol publication [ 50 ].
Rapid reviews can be used to summarize the evidence, but they involve methodological sacrifices and simplifications to produce information promptly, with inconsistent methodological approaches [ 51 ]. However, rapid reviews are justified in times of public health emergencies, and even Cochrane has resorted to publishing rapid reviews in response to the COVID-19 crisis [ 52 ]. Rapid reviews were eligible for inclusion in this overview, but only one of the 18 reviews included in this study was labeled as a rapid review.
Ideally, COVID-19 evidence would be continually summarized in a series of high-quality living systematic reviews, types of evidence synthesis defined as “ a systematic review which is continually updated, incorporating relevant new evidence as it becomes available ” [ 53 ]. However, conducting living systematic reviews requires considerable resources, calling into question the sustainability of such evidence synthesis over long periods [ 54 ].
Research reports about COVID-19 will contribute to research waste if they are poorly designed, poorly reported, or simply not necessary. In principle, systematic reviews should help reduce research waste as they usually provide recommendations for further research that is needed or may advise that sufficient evidence exists on a particular topic [ 55 ]. However, systematic reviews can also contribute to growing research waste when they are not needed, or poorly conducted and reported. Our present study clearly shows that most of the systematic reviews that were published early on in the COVID-19 pandemic could be categorized as research waste, as our confidence in their results is critically low.
Our study has some limitations. One is that for AMSTAR 2 assessment we relied on information available in publications; we did not attempt to contact study authors for clarifications or additional data. In three reviews, the methodological quality appraisal was challenging because they were published as letters, or labeled as rapid communications. As a result, various details about their review process were not included, leading to AMSTAR 2 questions being answered as “not reported”, resulting in low confidence scores. Full manuscripts might have provided additional information that could have led to higher confidence in the results. In other words, low scores could reflect incomplete reporting, not necessarily low-quality review methods. To make their review available more rapidly and more concisely, the authors may have omitted methodological details. A general issue during a crisis is that speed and completeness must be balanced. However, maintaining high standards requires proper resourcing and commitment to ensure that the users of systematic reviews can have high confidence in the results.
Furthermore, we used adjusted AMSTAR 2 scoring, as the tool was designed for critical appraisal of reviews of interventions. Some reviews may have received lower scores than actually warranted in spite of these adjustments.
Another limitation of our study may be the inclusion of multiple overlapping reviews, as some included reviews included the same primary studies. According to the Cochrane Handbook, including overlapping reviews may be appropriate when the review’s aim is “ to present and describe the current body of systematic review evidence on a topic ” [ 12 ], which was our aim. To avoid bias with summarizing evidence from overlapping reviews, we presented the forest plots without summary estimates. The forest plots serve to inform readers about the effect sizes for outcomes that were reported in each review.
Several authors from this study have contributed to one of the reviews identified [ 25 ]. To reduce the risk of any bias, two authors who did not co-author the review in question initially assessed its quality and limitations.
Finally, we note that the systematic reviews included in our overview may have had issues that our analysis did not identify because we did not analyze their primary studies to verify the accuracy of the data and information they presented. We give two examples to substantiate this possibility. Lovato et al. wrote a commentary on the review of Sun et al. [ 41 ], in which they criticized the authors’ conclusion that sore throat is rare in COVID-19 patients [ 56 ]. Lovato et al. highlighted that multiple studies included in Sun et al. did not accurately describe participants’ clinical presentations, warning that only three studies clearly reported data on sore throat [ 56 ].
In another example, Leung [ 57 ] warned about the review of Li, L.Q. et al. [ 29 ]: “ it is possible that this statistic was computed using overlapped samples, therefore some patients were double counted ”. Li et al. responded to Leung that it is uncertain whether the data overlapped, as they used data from published articles and did not have access to the original data; they also reported that they requested original data and that they plan to re-do their analyses once they receive them; they also urged readers to treat the data with caution [ 58 ]. This points to the evolving nature of evidence during a crisis.
Our study’s strength is that this overview adds to the current knowledge by providing a comprehensive summary of all the evidence synthesis about COVID-19 available early after the onset of the pandemic. This overview followed strict methodological criteria, including a comprehensive and sensitive search strategy and a standard tool for methodological appraisal of systematic reviews.
In conclusion, in this overview of systematic reviews, we analyzed evidence from the first 18 systematic reviews that were published after the emergence of COVID-19. However, confidence in the results of all the reviews was “critically low”. Thus, systematic reviews that were published early on in the pandemic could be categorized as research waste. Even during public health emergencies, studies and systematic reviews should adhere to established methodological standards to provide patients, clinicians, and decision-makers trustworthy evidence.
Availability of data and materials
All data collected and analyzed within this study are available from the corresponding author on reasonable request.
World Health Organization. Timeline - COVID-19: Available at: https://www.who.int/news/item/29-06-2020-covidtimeline . Accessed 1 June 2021.
COVID-19 Dashboard by the Center for Systems Science and Engineering (CSSE) at Johns Hopkins University (JHU). Available at: https://coronavirus.jhu.edu/map.html . Accessed 1 June 2021.
Anzai A, Kobayashi T, Linton NM, Kinoshita R, Hayashi K, Suzuki A, et al. Assessing the Impact of Reduced Travel on Exportation Dynamics of Novel Coronavirus Infection (COVID-19). J Clin Med. 2020;9(2):601.
Chinazzi M, Davis JT, Ajelli M, Gioannini C, Litvinova M, Merler S, et al. The effect of travel restrictions on the spread of the 2019 novel coronavirus (COVID-19) outbreak. Science. 2020;368(6489):395–400. https://doi.org/10.1126/science.aba9757 .
Article CAS PubMed PubMed Central Google Scholar
Fidahic M, Nujic D, Runjic R, Civljak M, Markotic F, Lovric Makaric Z, et al. Research methodology and characteristics of journal articles with original data, preprint articles and registered clinical trial protocols about COVID-19. BMC Med Res Methodol. 2020;20(1):161. https://doi.org/10.1186/s12874-020-01047-2 .
EPPI Centre . COVID-19: a living systematic map of the evidence. Available at: http://eppi.ioe.ac.uk/cms/Projects/DepartmentofHealthandSocialCare/Publishedreviews/COVID-19Livingsystematicmapoftheevidence/tabid/3765/Default.aspx . Accessed 1 June 2021.
NCBI SARS-CoV-2 Resources. Available at: https://www.ncbi.nlm.nih.gov/sars-cov-2/ . Accessed 1 June 2021.
Gustot T. Quality and reproducibility during the COVID-19 pandemic. JHEP Rep. 2020;2(4):100141. https://doi.org/10.1016/j.jhepr.2020.100141 .
Article PubMed PubMed Central Google Scholar
Kodvanj, I., et al., Publishing of COVID-19 Preprints in Peer-reviewed Journals, Preprinting Trends, Public Discussion and Quality Issues. Preprint article. bioRxiv 2020.11.23.394577; doi: https://doi.org/10.1101/2020.11.23.394577 .
Dobler CC. Poor quality research and clinical practice during COVID-19. Breathe (Sheff). 2020;16(2):200112. https://doi.org/10.1183/20734735.0112-2020 .
Article Google Scholar
Bastian H, Glasziou P, Chalmers I. Seventy-five trials and eleven systematic reviews a day: how will we ever keep up? PLoS Med. 2010;7(9):e1000326. https://doi.org/10.1371/journal.pmed.1000326 .
Lunny C, Brennan SE, McDonald S, McKenzie JE. Toward a comprehensive evidence map of overview of systematic review methods: paper 1-purpose, eligibility, search and data extraction. Syst Rev. 2017;6(1):231. https://doi.org/10.1186/s13643-017-0617-1 .
Pollock M, Fernandes RM, Becker LA, Pieper D, Hartling L. Chapter V: Overviews of Reviews. In: Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA (editors). Cochrane Handbook for Systematic Reviews of Interventions version 6.1 (updated September 2020). Cochrane. 2020. Available from www.training.cochrane.org/handbook .
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al. Cochrane handbook for systematic reviews of interventions version 6.1 (updated September 2020). Cochrane. 2020; Available from www.training.cochrane.org/handbook .
Pollock M, Fernandes RM, Newton AS, Scott SD, Hartling L. The impact of different inclusion decisions on the comprehensiveness and complexity of overviews of reviews of healthcare interventions. Syst Rev. 2019;8(1):18. https://doi.org/10.1186/s13643-018-0914-3 .
Pollock M, Fernandes RM, Newton AS, Scott SD, Hartling L. A decision tool to help researchers make decisions about including systematic reviews in overviews of reviews of healthcare interventions. Syst Rev. 2019;8(1):29. https://doi.org/10.1186/s13643-018-0768-8 .
Hunt H, Pollock A, Campbell P, Estcourt L, Brunton G. An introduction to overviews of reviews: planning a relevant research question and objective for an overview. Syst Rev. 2018;7(1):39. https://doi.org/10.1186/s13643-018-0695-8 .
Pollock M, Fernandes RM, Pieper D, Tricco AC, Gates M, Gates A, et al. Preferred reporting items for overviews of reviews (PRIOR): a protocol for development of a reporting guideline for overviews of reviews of healthcare interventions. Syst Rev. 2019;8(1):335. https://doi.org/10.1186/s13643-019-1252-9 .
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Open Med. 2009;3(3):e123–30.
Krnic Martinic M, Pieper D, Glatt A, Puljak L. Definition of a systematic review used in overviews of systematic reviews, meta-epidemiological studies and textbooks. BMC Med Res Methodol. 2019;19(1):203. https://doi.org/10.1186/s12874-019-0855-0 .
Puljak L. If there is only one author or only one database was searched, a study should not be called a systematic review. J Clin Epidemiol. 2017;91:4–5. https://doi.org/10.1016/j.jclinepi.2017.08.002 .
Article PubMed Google Scholar
Gates M, Gates A, Guitard S, Pollock M, Hartling L. Guidance for overviews of reviews continues to accumulate, but important challenges remain: a scoping review. Syst Rev. 2020;9(1):254. https://doi.org/10.1186/s13643-020-01509-0 .
Covidence - systematic review software. Available at: https://www.covidence.org/ . Accessed 1 June 2021.
Shea BJ, Reeves BC, Wells G, Thuku M, Hamel C, Moran J, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. 2017;358:j4008.
Borges do Nascimento IJ, et al. Novel Coronavirus Infection (COVID-19) in Humans: A Scoping Review and Meta-Analysis. J Clin Med. 2020;9(4):941.
Article PubMed Central Google Scholar
Adhikari SP, Meng S, Wu YJ, Mao YP, Ye RX, Wang QZ, et al. Epidemiology, causes, clinical manifestation and diagnosis, prevention and control of coronavirus disease (COVID-19) during the early outbreak period: a scoping review. Infect Dis Poverty. 2020;9(1):29. https://doi.org/10.1186/s40249-020-00646-x .
Cortegiani A, Ingoglia G, Ippolito M, Giarratano A, Einav S. A systematic review on the efficacy and safety of chloroquine for the treatment of COVID-19. J Crit Care. 2020;57:279–83. https://doi.org/10.1016/j.jcrc.2020.03.005 .
Li B, Yang J, Zhao F, Zhi L, Wang X, Liu L, et al. Prevalence and impact of cardiovascular metabolic diseases on COVID-19 in China. Clin Res Cardiol. 2020;109(5):531–8. https://doi.org/10.1007/s00392-020-01626-9 .
Article CAS PubMed Google Scholar
Li LQ, Huang T, Wang YQ, Wang ZP, Liang Y, Huang TB, et al. COVID-19 patients’ clinical characteristics, discharge rate, and fatality rate of meta-analysis. J Med Virol. 2020;92(6):577–83. https://doi.org/10.1002/jmv.25757 .
Lippi G, Lavie CJ, Sanchis-Gomar F. Cardiac troponin I in patients with coronavirus disease 2019 (COVID-19): evidence from a meta-analysis. Prog Cardiovasc Dis. 2020;63(3):390–1. https://doi.org/10.1016/j.pcad.2020.03.001 .
Lippi G, Henry BM. Active smoking is not associated with severity of coronavirus disease 2019 (COVID-19). Eur J Intern Med. 2020;75:107–8. https://doi.org/10.1016/j.ejim.2020.03.014 .
Lippi G, Plebani M. Procalcitonin in patients with severe coronavirus disease 2019 (COVID-19): a meta-analysis. Clin Chim Acta. 2020;505:190–1. https://doi.org/10.1016/j.cca.2020.03.004 .
Lippi G, Plebani M, Henry BM. Thrombocytopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: a meta-analysis. Clin Chim Acta. 2020;506:145–8. https://doi.org/10.1016/j.cca.2020.03.022 .
Ludvigsson JF. Systematic review of COVID-19 in children shows milder cases and a better prognosis than adults. Acta Paediatr. 2020;109(6):1088–95. https://doi.org/10.1111/apa.15270 .
Lupia T, Scabini S, Mornese Pinna S, di Perri G, de Rosa FG, Corcione S. 2019 novel coronavirus (2019-nCoV) outbreak: a new challenge. J Glob Antimicrob Resist. 2020;21:22–7. https://doi.org/10.1016/j.jgar.2020.02.021 .
Marasinghe, K.M., A systematic review investigating the effectiveness of face mask use in limiting the spread of COVID-19 among medically not diagnosed individuals: shedding light on current recommendations provided to individuals not medically diagnosed with COVID-19. Research Square. Preprint article. doi : https://doi.org/10.21203/rs.3.rs-16701/v1 . 2020 .
Mullins E, Evans D, Viner RM, O’Brien P, Morris E. Coronavirus in pregnancy and delivery: rapid review. Ultrasound Obstet Gynecol. 2020;55(5):586–92. https://doi.org/10.1002/uog.22014 .
Pang J, Wang MX, Ang IYH, Tan SHX, Lewis RF, Chen JIP, et al. Potential Rapid Diagnostics, Vaccine and Therapeutics for 2019 Novel coronavirus (2019-nCoV): a systematic review. J Clin Med. 2020;9(3):623.
Rodriguez-Morales AJ, Cardona-Ospina JA, Gutiérrez-Ocampo E, Villamizar-Peña R, Holguin-Rivera Y, Escalera-Antezana JP, et al. Clinical, laboratory and imaging features of COVID-19: a systematic review and meta-analysis. Travel Med Infect Dis. 2020;34:101623. https://doi.org/10.1016/j.tmaid.2020.101623 .
Salehi S, Abedi A, Balakrishnan S, Gholamrezanezhad A. Coronavirus disease 2019 (COVID-19): a systematic review of imaging findings in 919 patients. AJR Am J Roentgenol. 2020;215(1):87–93. https://doi.org/10.2214/AJR.20.23034 .
Sun P, Qie S, Liu Z, Ren J, Li K, Xi J. Clinical characteristics of hospitalized patients with SARS-CoV-2 infection: a single arm meta-analysis. J Med Virol. 2020;92(6):612–7. https://doi.org/10.1002/jmv.25735 .
Yang J, Zheng Y, Gou X, Pu K, Chen Z, Guo Q, et al. Prevalence of comorbidities and its effects in patients infected with SARS-CoV-2: a systematic review and meta-analysis. Int J Infect Dis. 2020;94:91–5. https://doi.org/10.1016/j.ijid.2020.03.017 .
Bassetti M, Vena A, Giacobbe DR. The novel Chinese coronavirus (2019-nCoV) infections: challenges for fighting the storm. Eur J Clin Investig. 2020;50(3):e13209. https://doi.org/10.1111/eci.13209 .
Article CAS Google Scholar
Hwang CS. Olfactory neuropathy in severe acute respiratory syndrome: report of a case. Acta Neurol Taiwanica. 2006;15(1):26–8.
Google Scholar
Suzuki M, Saito K, Min WP, Vladau C, Toida K, Itoh H, et al. Identification of viruses in patients with postviral olfactory dysfunction. Laryngoscope. 2007;117(2):272–7. https://doi.org/10.1097/01.mlg.0000249922.37381.1e .
Rajgor DD, Lee MH, Archuleta S, Bagdasarian N, Quek SC. The many estimates of the COVID-19 case fatality rate. Lancet Infect Dis. 2020;20(7):776–7. https://doi.org/10.1016/S1473-3099(20)30244-9 .
Wolkewitz M, Puljak L. Methodological challenges of analysing COVID-19 data during the pandemic. BMC Med Res Methodol. 2020;20(1):81. https://doi.org/10.1186/s12874-020-00972-6 .
Rombey T, Lochner V, Puljak L, Könsgen N, Mathes T, Pieper D. Epidemiology and reporting characteristics of non-Cochrane updates of systematic reviews: a cross-sectional study. Res Synth Methods. 2020;11(3):471–83. https://doi.org/10.1002/jrsm.1409 .
Runjic E, Rombey T, Pieper D, Puljak L. Half of systematic reviews about pain registered in PROSPERO were not published and the majority had inaccurate status. J Clin Epidemiol. 2019;116:114–21. https://doi.org/10.1016/j.jclinepi.2019.08.010 .
Runjic E, Behmen D, Pieper D, Mathes T, Tricco AC, Moher D, et al. Following Cochrane review protocols to completion 10 years later: a retrospective cohort study and author survey. J Clin Epidemiol. 2019;111:41–8. https://doi.org/10.1016/j.jclinepi.2019.03.006 .
Tricco AC, Antony J, Zarin W, Strifler L, Ghassemi M, Ivory J, et al. A scoping review of rapid review methods. BMC Med. 2015;13(1):224. https://doi.org/10.1186/s12916-015-0465-6 .
COVID-19 Rapid Reviews: Cochrane’s response so far. Available at: https://training.cochrane.org/resource/covid-19-rapid-reviews-cochrane-response-so-far . Accessed 1 June 2021.
Cochrane. Living systematic reviews. Available at: https://community.cochrane.org/review-production/production-resources/living-systematic-reviews . Accessed 1 June 2021.
Millard T, Synnot A, Elliott J, Green S, McDonald S, Turner T. Feasibility and acceptability of living systematic reviews: results from a mixed-methods evaluation. Syst Rev. 2019;8(1):325. https://doi.org/10.1186/s13643-019-1248-5 .
Babic A, Poklepovic Pericic T, Pieper D, Puljak L. How to decide whether a systematic review is stable and not in need of updating: analysis of Cochrane reviews. Res Synth Methods. 2020;11(6):884–90. https://doi.org/10.1002/jrsm.1451 .
Lovato A, Rossettini G, de Filippis C. Sore throat in COVID-19: comment on “clinical characteristics of hospitalized patients with SARS-CoV-2 infection: a single arm meta-analysis”. J Med Virol. 2020;92(7):714–5. https://doi.org/10.1002/jmv.25815 .
Leung C. Comment on Li et al: COVID-19 patients’ clinical characteristics, discharge rate, and fatality rate of meta-analysis. J Med Virol. 2020;92(9):1431–2. https://doi.org/10.1002/jmv.25912 .
Li LQ, Huang T, Wang YQ, Wang ZP, Liang Y, Huang TB, et al. Response to Char’s comment: comment on Li et al: COVID-19 patients’ clinical characteristics, discharge rate, and fatality rate of meta-analysis. J Med Virol. 2020;92(9):1433. https://doi.org/10.1002/jmv.25924 .
Download references
Acknowledgments
We thank Catherine Henderson DPhil from Swanscoe Communications for pro bono medical writing and editing support. We acknowledge support from the Covidence Team, specifically Anneliese Arno. We thank the whole International Network of Coronavirus Disease 2019 (InterNetCOVID-19) for their commitment and involvement. Members of the InterNetCOVID-19 are listed in Additional file 6 . We thank Pavel Cerny and Roger Crosthwaite for guiding the team supervisor (IJBN) on human resources management.
This research received no external funding.
Author information
Authors and affiliations.
University Hospital and School of Medicine, Universidade Federal de Minas Gerais, Belo Horizonte, Minas Gerais, Brazil
Israel Júnior Borges do Nascimento & Milena Soriano Marcolino
Medical College of Wisconsin, Milwaukee, WI, USA
Israel Júnior Borges do Nascimento
Helene Fuld Health Trust National Institute for Evidence-based Practice in Nursing and Healthcare, College of Nursing, The Ohio State University, Columbus, OH, USA
Dónal P. O’Mathúna
School of Nursing, Psychotherapy and Community Health, Dublin City University, Dublin, Ireland
Department of Anesthesiology, Intensive Care and Pain Medicine, University of Münster, Münster, Germany
Thilo Caspar von Groote
Department of Sport and Health Science, Technische Universität München, Munich, Germany
Hebatullah Mohamed Abdulazeem
School of Health Sciences, Faculty of Health and Medicine, The University of Newcastle, Callaghan, Australia
Ishanka Weerasekara
Department of Physiotherapy, Faculty of Allied Health Sciences, University of Peradeniya, Peradeniya, Sri Lanka
Cochrane Croatia, University of Split, School of Medicine, Split, Croatia
Ana Marusic, Irena Zakarija-Grkovic & Tina Poklepovic Pericic
Center for Evidence-Based Medicine and Health Care, Catholic University of Croatia, Ilica 242, 10000, Zagreb, Croatia
Livia Puljak
Cochrane Brazil, Evidence-Based Health Program, Universidade Federal de São Paulo, São Paulo, Brazil
Vinicius Tassoni Civile & Alvaro Nagib Atallah
Yorkville University, Fredericton, New Brunswick, Canada
Santino Filoso
Laboratory for Industrial and Applied Mathematics (LIAM), Department of Mathematics and Statistics, York University, Toronto, Ontario, Canada
Nicola Luigi Bragazzi
You can also search for this author in PubMed Google Scholar
Contributions
IJBN conceived the research idea and worked as a project coordinator. DPOM, TCVG, HMA, IW, AM, LP, VTC, IZG, TPP, ANA, SF, NLB and MSM were involved in data curation, formal analysis, investigation, methodology, and initial draft writing. All authors revised the manuscript critically for the content. The author(s) read and approved the final manuscript.
Corresponding author
Correspondence to Livia Puljak .
Ethics declarations
Ethics approval and consent to participate.
Not required as data was based on published studies.
Consent for publication
Not applicable.
Competing interests
The authors declare no conflict of interest.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: appendix 1..
Search strategies used in the study.
Additional file 2: Appendix 2.
Adjusted scoring of AMSTAR 2 used in this study for systematic reviews of studies that did not analyze interventions.
Additional file 3: Appendix 3.
List of excluded studies, with reasons.
Additional file 4: Appendix 4.
Table of overlapping studies, containing the list of primary studies included, their visual overlap in individual systematic reviews, and the number in how many reviews each primary study was included.
Additional file 5: Appendix 5.
A detailed explanation of AMSTAR scoring for each item in each review.
Additional file 6: Appendix 6.
List of members and affiliates of International Network of Coronavirus Disease 2019 (InterNetCOVID-19).
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Borges do Nascimento, I.J., O’Mathúna, D.P., von Groote, T.C. et al. Coronavirus disease (COVID-19) pandemic: an overview of systematic reviews. BMC Infect Dis 21 , 525 (2021). https://doi.org/10.1186/s12879-021-06214-4
Download citation
Received : 12 April 2020
Accepted : 19 May 2021
Published : 04 June 2021
DOI : https://doi.org/10.1186/s12879-021-06214-4
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Coronavirus
- Evidence-based medicine
- Infectious diseases
BMC Infectious Diseases
ISSN: 1471-2334
- Submission enquiries: [email protected]
- General enquiries: [email protected]
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- My Account Login
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
- Open access
- Published: 11 February 2021
Methodological quality of COVID-19 clinical research
- Richard G. Jung ORCID: orcid.org/0000-0002-8570-6736 1 , 2 , 3 na1 ,
- Pietro Di Santo 1 , 2 , 4 , 5 na1 ,
- Cole Clifford 6 ,
- Graeme Prosperi-Porta 7 ,
- Stephanie Skanes 6 ,
- Annie Hung 8 ,
- Simon Parlow 4 ,
- Sarah Visintini ORCID: orcid.org/0000-0001-6966-1753 9 ,
- F. Daniel Ramirez ORCID: orcid.org/0000-0002-4350-1652 1 , 4 , 10 , 11 ,
- Trevor Simard 1 , 2 , 3 , 4 , 12 &
- Benjamin Hibbert ORCID: orcid.org/0000-0003-0906-1363 2 , 3 , 4
Nature Communications volume 12 , Article number: 943 ( 2021 ) Cite this article
13k Accesses
93 Citations
238 Altmetric
Metrics details
- Infectious diseases
- Public health
The COVID-19 pandemic began in early 2020 with major health consequences. While a need to disseminate information to the medical community and general public was paramount, concerns have been raised regarding the scientific rigor in published reports. We performed a systematic review to evaluate the methodological quality of currently available COVID-19 studies compared to historical controls. A total of 9895 titles and abstracts were screened and 686 COVID-19 articles were included in the final analysis. Comparative analysis of COVID-19 to historical articles reveals a shorter time to acceptance (13.0[IQR, 5.0–25.0] days vs. 110.0[IQR, 71.0–156.0] days in COVID-19 and control articles, respectively; p < 0.0001). Furthermore, methodological quality scores are lower in COVID-19 articles across all study designs. COVID-19 clinical studies have a shorter time to publication and have lower methodological quality scores than control studies in the same journal. These studies should be revisited with the emergence of stronger evidence.
Similar content being viewed by others
Clinical presentations, laboratory and radiological findings, and treatments for 11,028 COVID-19 patients: a systematic review and meta-analysis
Improving clinical paediatric research and learning from COVID-19: recommendations by the Conect4Children expert advice group
The effect of influenza vaccine in reducing the severity of clinical outcomes in patients with COVID-19: a systematic review and meta-analysis
Introduction.
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic spread globally in early 2020 with substantial health and economic consequences. This was associated with an exponential increase in scientific publications related to the coronavirus disease 2019 (COVID-19) in order to rapidly elucidate the natural history and identify diagnostic and therapeutic tools 1 .
While a need to rapidly disseminate information to the medical community, governmental agencies, and general public was paramount—major concerns have been raised regarding the scientific rigor in the literature 2 . Poorly conducted studies may originate from failure at any of the four consecutive research stages: (1) choice of research question relevant to patient care, (2) quality of research design 3 , (3) adequacy of publication, and (4) quality of research reports. Furthermore, evidence-based medicine relies on a hierarchy of evidence, ranging from the highest level of randomized controlled trials (RCT) to the lowest level of case series and case reports 4 .
Given the implications for clinical care, policy decision making, and concerns regarding methodological and peer-review standards for COVID-19 research 5 , we performed a formal evaluation of the methodological quality of published COVID-19 literature. Specifically, we undertook a systematic review to identify COVID-19 clinical literature and matched them to historical controls to formally evaluate the following: (1) the methodological quality of COVID-19 studies using established quality tools and checklists, (2) the methodological quality of COVID-19 studies, stratified by median time to acceptance, geographical regions, and journal impact factor and (3) a comparison of COVID-19 methodological quality to matched controls.
Herein, we show that COVID-19 articles are associated with lower methodological quality scores. Moreover, in a matched cohort analysis with control articles from the same journal, we reveal that COVID-19 articles are associated with lower quality scores and shorter time from submission to acceptance. Ultimately, COVID-19 clinical studies should be revisited with the emergence of stronger evidence.

Article selection
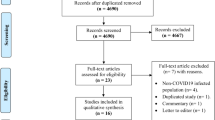
A total of 14787 COVID-19 papers were identified as of May 14, 2020 and 4892 duplicate articles were removed. In total, 9895 titles and abstracts were screened, and 9101 articles were excluded due to the study being pre-clinical in nature, case report, case series <5 patients, in a language other than English, reviews (including systematic reviews), study protocols or methods, and other coronavirus variants with an overall inter-rater study inclusion agreement of 96.7% ( κ = 0.81; 95% CI, 0.79–0.83). A total number of 794 full texts were reviewed for eligibility. Over 108 articles were excluded for ineligible study design or publication type (such as letter to the editors, editorials, case reports or case series <5 patients), wrong patient population, non-English language, duplicate articles, wrong outcomes and publication in a non-peer-reviewed journal. Ultimately, 686 articles were identified with an inter-rater agreement of 86.5% ( κ = 0.68; 95% CI, 0.67–0.70) (Fig. 1 ).
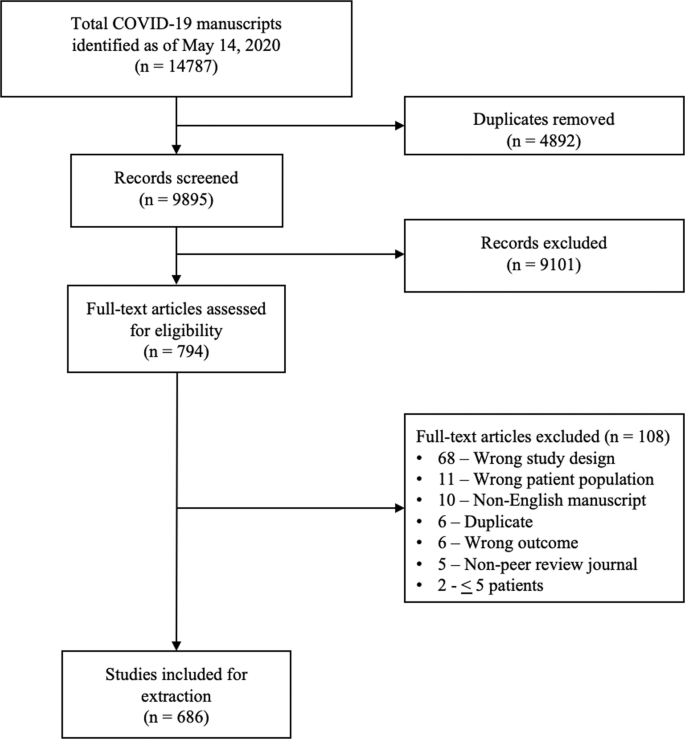
A total of 14787 articles were identified and 4892 duplicate articles were removed. Overall, 9895 articles were screened by title and abstract leaving 794 articles for full-text screening. Over 108 articles were excluded, leaving a total of 686 articles that underwent methodological quality assessment.
COVID-19 literature methodological quality
Most studies originated from Asia/Oceania with 469 (68.4%) studies followed by Europe with 139 (20.3%) studies, and the Americas with 78 (11.4%) studies. Of included studies, 380 (55.4%) were case series, 199 (29.0%) were cohort, 63 (9.2%) were diagnostic, 38 (5.5%) were case–control, and 6 (0.9%) were RCTs. Most studies (590, 86.0%) were retrospective in nature, 620 (90.4%) reported the sex of patients, and 7 (2.3%) studies excluding case series calculated their sample size a priori. The method of SARS-CoV-2 diagnosis was reported in 558 studies (81.3%) and ethics approval was obtained in 556 studies (81.0%). Finally, journal impact factor of COVID-19 manuscripts was 4.7 (IQR, 2.9–7.6) with a time to acceptance of 13.0 (IQR, 5.0–25.0) days (Table 1 ).
Overall, when COVID-19 articles were stratified by study design, a mean case series score (out of 5) (SD) of 3.3 (1.1), mean NOS cohort study score (out of 8) of 5.8 (1.5), mean NOS case–control study score (out of 8) of 5.5 (1.9), and low bias present in 4 (6.4%) diagnostic studies was observed (Table 2 and Fig. 2 ). Furthermore, in the 6 RCTs in the COVID-19 literature, there was a high risk of bias with little consideration for sequence generation, allocation concealment, blinding, incomplete outcome data, and selective outcome reporting (Table 2 ).
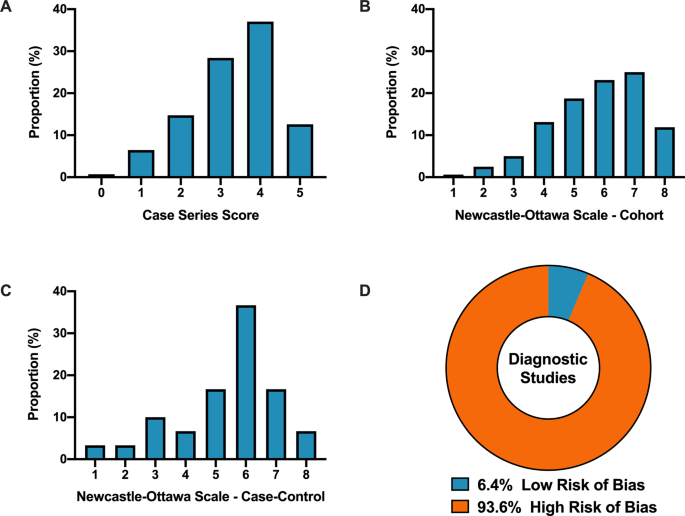
A Distribution of COVID-19 case series studies scored using the Murad tool ( n = 380). B Distribution of COVID-19 cohort studies scored using the Newcastle–Ottawa Scale ( n = 199). C Distribution of COVID-19 case–control studies scored using the Newcastle–Ottawa Scale ( n = 38). D Distribution of COVID-19 diagnostic studies scored using the QUADAS-2 tool ( n = 63). In panel D , blue represents low risk of bias and orange represents high risk of bias.
For secondary outcomes, rapid time from submission to acceptance (stratified by median time of acceptance of <13.0 days) was associated with lower methodological quality scores for case series and cohort study designs but not for case–control nor diagnostic studies (Fig. 3A–D ). Low journal impact factor (<10) was associated with lower methodological quality scores for case series, cohort, and case–control designs (Fig. 3E–H ). Finally, studies originating from different geographical regions had no differences in methodological quality scores with the exception of cohort studies (Fig. 3I–L ). When dichotomized by high vs. low methodological quality scores, a similar trend was observed with rapid time from submission to acceptance (34.4% vs. 46.3%, p = 0.01, Supplementary Fig. 1B ), low impact factor journals (<10) was associated with lower methodological quality score (38.8% vs. 68.0%, p < 0.0001, Supplementary Fig. 1C ). Finally, studies originating in either Americas or Asia/Oceania was associated with higher methodological quality scores than Europe (Supplementary Fig. 1D ).
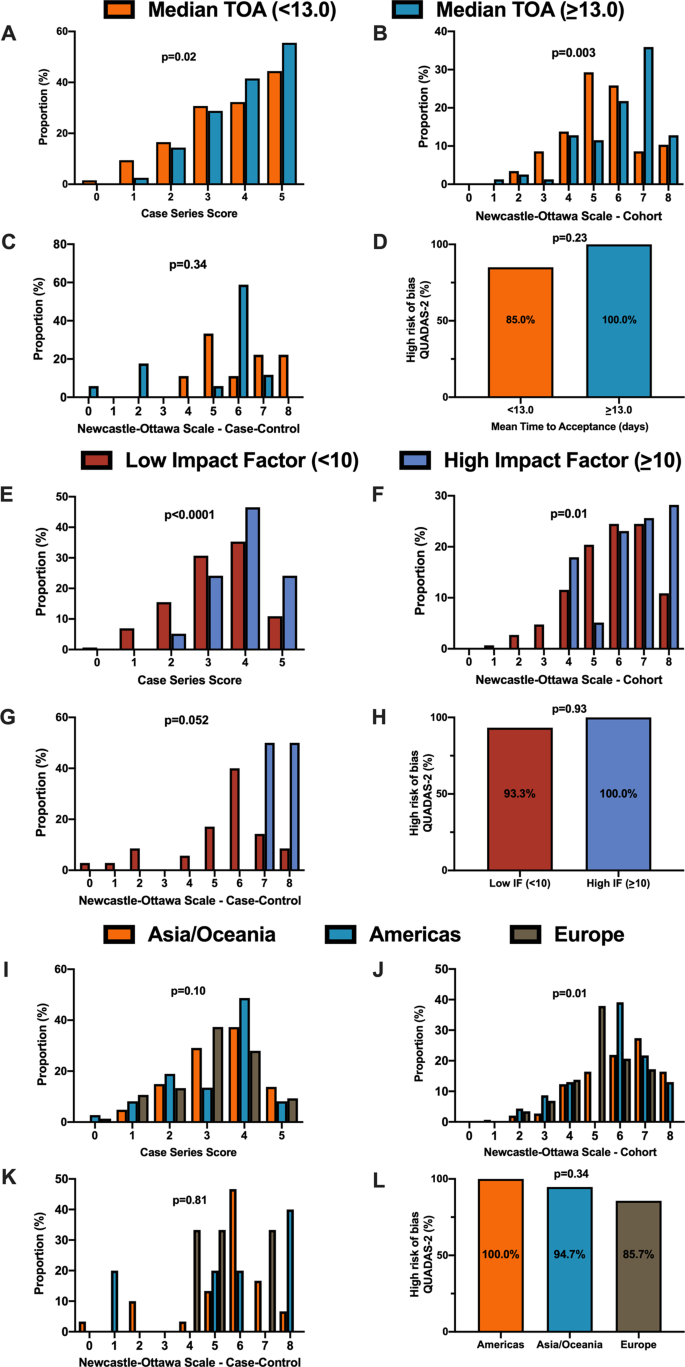
A When stratified by time of acceptance (13.0 days), increased time of acceptance was associated with higher case series score ( n = 186 for <13 days and n = 193 for >=13 days; p = 0.02). B Increased time of acceptance was associated with higher NOS cohort score ( n = 112 for <13 days and n = 144 for >=13 days; p = 0.003). C No difference in time of acceptance and case–control score was observed ( n = 18 for <13 days and n = 27 for >=13 days; p = 0.34). D No difference in time of acceptance and diagnostic risk of bias (QUADAS-2) was observed ( n = 43 for <13 days and n = 33 for >=13 days; p = 0.23). E When stratified by impact factor (IF ≥10), high IF was associated with higher case series score ( n = 466 for low IF and n = 60 for high IF; p < 0.0001). F High IF was associated with higher NOS cohort score ( n = 262 for low IF and n = 68 for high IF; p = 0.01). G No difference in IF and case–control score was observed ( n = 62 for low IF and n = 2 for high IF; p = 0.052). H No difference in IF and QUADAS-2 was observed ( n = 101 for low IF and n = 2 for high IF; p = 0.93). I When stratified by geographical region, no difference in geographical region and case series score was observed ( n = 276 Asia/Oceania, n = 135 Americas, and n = 143 Europe/Africa; p = 0.10). J Geographical region was associated with differences in cohort score ( n = 177 Asia/Oceania, n = 81 Americas, and n = 89 Europe/Africa; p = 0.01). K No difference in geographical region and case–control score was observed ( n = 37 Asia/Oceania, n = 13 Americas, and n = 14 Europe/Africa; p = 0.81). L No difference in geographical region and QUADAS-2 was observed ( n = 49 Asia/Oceania, n = 28 Americas, and n = 28 Europe/Africa; p = 0.34). In panels A – D , orange represents lower median time of acceptance and blue represents high median time of acceptance. In panels E – H , red is low impact factor and blue is high impact factor. In panels I – L , orange represents Asia/Oceania, blue represents Americas, and brown represents Europe. Differences in distributions were analysed by two-sided Kruskal–Wallis test. Differences in diagnostic risk of bias were quantified by Chi-squares test. p < 0.05 was considered statistically significant.
Methodological quality score differences in COVID-19 versus historical control
We matched 539 historical control articles to COVID-19 articles from the same journal with identical study designs in the previous year for a final analysis of 1078 articles (Table 1 ). Overall, 554 (51.4%) case series, 348 (32.3%) cohort, 64 (5.9%) case–control, 106 (9.8%) diagnostic and 6 (0.6%) RCTs were identified from the 1078 total articles. Differences exist between COVID-19 and historical control articles in geographical region of publication, retrospective study design, and sample size calculation (Table 1 ). Time of acceptance was 13.0 (IQR, 5.0–25.0) days in COVID-19 articles vs. 110.0 (IQR, 71.0–156.0) days in control articles (Table 1 and Fig. 4A , p < 0.0001). Case-series methodological quality score was lower in COVID-19 articles compared to the historical control (3.3 (1.1) vs. 4.3 (0.8); n = 554; p < 0.0001; Table 2 and Fig. 4B ). Furthermore, NOS score was lower in COVID-19 cohort studies (5.8 (1.6) vs. 7.1 (1.0); n = 348; p < 0.0001; Table 2 and Fig. 4C ) and case–control studies (5.4 (1.9) vs. 6.6 (1.0); n = 64; p = 0.003; Table 2 and Fig. 4D ). Finally, lower risk of bias in diagnostic studies was in 12 COVID-19 articles (23%; n = 53) compared to 24 control articles (45%; n = 53; p = 0.02; Table 2 and Fig. 4E ). A similar trend was observed between COVID-19 and historical control articles when dichotomized by good vs. low methodological quality scores (Supplementary Fig. 2 ).
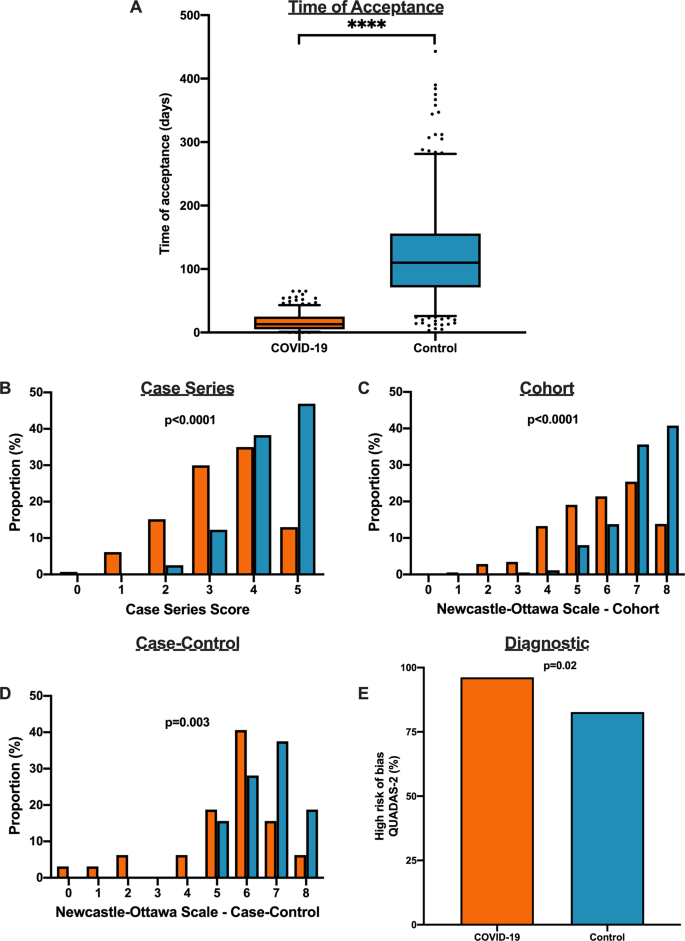
A Time to acceptance was reduced in COVID-19 articles compared to control articles (13.0 [IQR, 5.0–25.0] days vs. 110.0 [IQR, 71.0–156.0] days, n = 347 for COVID-19 and n = 414 for controls; p < 0.0001). B When compared to historical control articles, COVID-19 articles were associated with lower case series score ( n = 277 for COVID-19 and n = 277 for controls; p < 0.0001). C COVID-19 articles were associated with lower NOS cohort score compared to historical control articles ( n = 174 for COVID-19 and n = 174 for controls; p < 0.0001). D COVID-19 articles were associated with lower NOS case–control score compared to historical control articles ( n = 32 for COVID-19 and n = 32 for controls; p = 0.003). E COVID-19 articles were associated with higher diagnostic risk of bias (QUADAS-2) compared to historical control articles ( n = 53 for COVID-19 and n = 53 for controls; p = 0.02). For panel A , boxplot captures 5, 25, 50, 75 and 95% from the first to last whisker. Orange represents COVID-19 articles and blue represents control articles. Two-sided Mann–Whitney U-test was conducted to evaluate differences in time to acceptance between COVID-19 and control articles. Differences in study quality scores were evaluated by two-sided Kruskal–Wallis test. Differences in diagnostic risk of bias were quantified by Chi-squares test. p < 0.05 was considered statistically significant.
In this systematic evaluation of methodological quality, COVID-19 clinical research was primarily observational in nature with modest methodological quality scores. Not only were the study designs low in the hierarchy of scientific evidence, we found that COVID-19 articles were associated with a lower methodological quality scores when published with a shorter time of publication and in lower impact factor journals. Furthermore, in a matched cohort analysis with historical control articles identified from the same journal of the same study design, we demonstrated that COVID-19 articles were associated with lower quality scores and shorter time from submission to acceptance.
The present study demonstrates comparative differences in methodological quality scores between COVID-19 literature and historical control articles. Overall, the accelerated publication of COVID-19 research was associated with lower study quality scores compared to previously published historical control studies. Our research highlights major differences in study quality between COVID-19 and control articles, possibly driven in part by a combination of more thorough editorial and/or peer-review process as suggested by the time to publication, and robust study design with questions which are pertinent for clinicians and patient management 3 , 6 , 7 , 8 , 9 , 10 , 11 .
In the early stages of the COVID-19 pandemic, we speculate that an urgent need for scientific data to inform clinical, social and economic decisions led to shorter time to publication and explosion in publication of COVID-19 studies in both traditional peer-reviewed journals and preprint servers 1 , 12 . The accelerated scientific process in the COVID-19 pandemic allowed a rapid understanding of natural history of COVID-19 symptomology and prognosis, identification of tools including RT-PCR to diagnose SARS-CoV-2 13 , and identification of potential therapeutic options such as tocilizumab and convalescent plasma which laid the foundation for future RCTs 14 , 15 , 16 . A delay in publication of COVID-19 articles due to a slower peer-review process may potentially delay dissemination of pertinent information against the pandemic. Despite concerns of slow peer review, major landmark trials (i.e. RECOVERY and ACTT-1 trial) 17 , 18 published their findings in preprint servers and media releases to allow for rapid dissemination. Importantly, the data obtained in these initial studies should be revisited as stronger data emerges as lower quality studies may fundamentally risk patient safety, resource allocation and future scientific research 19 .
Unfortunately, poor evidence begets poor clinical decisions 20 . Furthermore, lower quality scientific evidence potentially undermines the public’s trust in science during this time and has been evident through misleading information and high-profile retractions 12 , 21 , 22 , 23 . For example, the benefits of hydroxychloroquine, which were touted early in the pandemic based on limited data, have subsequently failed to be replicated in multiple observational studies and RCTs 5 , 24 , 25 , 26 , 27 , 28 , 29 , 30 . One poorly designed study combined with rapid publication led to considerable investment of both the scientific and medical community—akin to quinine being sold to the public as a miracle drug during the 1918 Spanish Influenza 31 , 32 . Moreover, as of June 30, 2020, ClinicalTrials.gov listed an astonishing 230 COVID-19 trials with hydroxychloroquine/plaquenil, and a recent living systematic review of observational studies and RCTs of hydroxychloroquine or chloroquine for COVID-19 demonstrated no evidence of benefit nor harm with concerns of severe methodological flaws in the included studies 33 .
Our study has important limitations. We evaluated the methodological quality of existing studies using established checklists and tools. While it is tempting to associate methodological quality scores with reproducibility or causal inferences of the intervention, it is not possible to ascertain the impact on the study design and conduct of research nor results or conclusions in the identified reports 34 . Second, although the methodological quality scales and checklists used for the manuscript are commonly used for quality assessment in systematic reviews and meta-analyses 35 , 36 , 37 , 38 , they can only assess the methodology without consideration for causal language and are prone to limitations 39 , 40 . Other tools such as the ROBINS-I and GRADE exist to evaluate methodological quality of identified manuscripts, although no consensus currently exists for critical appraisal of non-randomized studies 41 , 42 , 43 . Furthermore, other considerations of quality such as sample size calculation, sex reporting or ethics approval are not considered in these quality scores. As such, the quality scores measured using these checklists only reflect the patient selection, comparability, diagnostic reference standard and methods to ascertain the outcome of the study. Third, the 1:1 ratio to identify our historical control articles may affect the precision estimates of our findings. Interestingly, a simulation of an increase from 1:1 to 1:4 control ratio tightened the precision estimates but did not significantly alter the point estimate 44 . Furthermore, the decision for 1:1 ratio in our study exists due to limitations of available historical control articles from the identical journal in the restricted time period combined with a large effect size and sample size in the analysis. Finally, our analysis includes early publications on COVID-19 and there is likely to be an improvement in quality of related studies and study design as the field matures and higher-quality studies. Accordingly, our findings are limited to the early body of research as it pertains to the pandemic and it is likely that over time research quality will improve over time.
In summary, the early body of peer-reviewed COVID-19 literature was composed primarily of observational studies that underwent shorter peer-review evaluation and were associated with lower methodological quality scores than comparable studies. COVID-19 clinical studies should be revisited with the emergence of stronger evidence.
A systematic literature search was conducted on May 14, 2020 (registered on June 3, 2020 at PROSPERO: CRD42020187318) and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses. Furthermore, the cohort study was reported according to the Strengthening The Reporting of Observational Studies in Epidemiology checklist. The data supporting the findings of this study is available as Supplementary Data 1 – 2 .
Data sources and searches
The search was created in MEDLINE by a medical librarian with expertise in systematic reviews (S.V.) using a combination of key terms and index headings related to COVID-19 and translated to the remaining bibliographic databases (Supplementary Tables 1 – 3 ). The searches were conducted in MEDLINE (Ovid MEDLINE(R) ALL 1946–), Embase (Ovid Embase Classic + Embase 1947–) and the Cochrane Central Register of Controlled Trials (from inception). Search results were limited to English-only publications, and a publication date limit of January 1, 2019 to present was applied. In addition, a Canadian Agency for Drugs and Technologies in Health search filter was applied in MEDLINE and Embase to remove animal studies, and commentary, newspaper article, editorial, letter and note publication types were also eliminated. Search results were exported to Covidence (Veritas Health Innovation, Melbourne, Australia) and duplicates were eliminated using the platform’s duplicate identification feature.
Study selection, data extraction and methodological quality assessment
We included all types of COVID-19 clinical studies, including case series, observational studies, diagnostic studies and RCTs. For diagnostic studies, the reference standard for COVID-19 diagnosis was defined as a nasopharyngeal swab followed by reverse transcriptase-polymerase chain reaction in order to detect SARS-CoV-2. We excluded studies that were exploratory or pre-clinical in nature (i.e. in vitro or animal studies), case reports or case series of <5 patients, studies published in a language other than English, reviews, methods or protocols, and other coronavirus variants such as the Middle East respiratory syndrome.
The review team consisted of trained research staff with expertise in systematic reviews and one trainee. Title and abstracts were evaluated by two independent reviewers using Covidence and all discrepancies were resolved by consensus. Articles that were selected for full review were independently evaluated by two reviewers for quality assessment using a standardized case report form following the completion of a training period where all reviewers were trained with the original manuscripts which derived the tools or checklists along with examples for what were deemed high scores 35 , 36 , 37 , 38 . Following this, reviewers completed thirty full-text extractions and the two reviewers had to reach consensus and the process was repeated for the remaining manuscripts independently. When two independent reviewers were not able reach consensus, a third reviewer (principal investigator) provided oversight in the process to resolve the conflicted scores.
First and corresponding author names, date of publication, title of manuscript and journal of publication were collected for all included full-text articles. Journal impact factor was obtained from the 2018 InCites Journal Citation Reports from Clarivate Analytics. Submission and acceptance dates were collected in manuscripts when available. Other information such as study type, prospective or retrospective study, sex reporting, sample size calculation, method of SARS-CoV-2 diagnosis and ethics approval was collected by the authors. Methodological quality assessment was conducted using the Newcastle–Ottawa Scale (NOS) for case–control and cohort studies 37 , QUADAS-2 tool for diagnostic studies 38 , Cochrane risk of bias for RCTs 35 and a score derived by Murad et al. for case series studies 36 .
Identification of historical control from identified COVID-19 articles
Following the completion of full-text extraction of COVID-19 articles, we obtained a historical control group by identifying reports matched in a 1:1 fashion. From the eligible COVID-19 article, historical controls were identified by searching the same journal in a systematic fashion by matching the same study design (“case series”, “cohort”, “case control” or “diagnostic”) starting in the journal edition 12 months prior to the COVID-19 article publication on the publisher website (i.e. COVID-19 article published on April 2020, going backwards to April 2019) and proceeding forward (or backward if a specific article type was not identified) in a temporal fashion until the first matched study was identified following abstract screening by two independent reviewers. If no comparison article was found by either reviewers, the corresponding COVID-19 article was excluded from the comparison analysis. Following the identification of the historical control, data extraction and quality assessment was conducted on the identified articles using the standardized case report forms by two independent reviewers and conflicts resolved by consensus. The full dataset has been made available as Supplementary Data 1 – 2 .
Data synthesis and statistical analysis
Continuous variables were reported as mean (SD) or median (IQR) as appropriate, and categorical variables were reported as proportions (%). Continuous variables were compared using Student t -test or Mann–Whitney U-test and categorical variables including quality scores were compared by χ 2 , Fisher’s exact test, or Kruskal–Wallis test.
The primary outcome of interest was to evaluate the methodological quality of COVID-19 clinical literature by study design using the Newcastle–Ottawa Scale (NOS) for case–control and cohort studies, QUADAS-2 tool for diagnostic studies 38 , Cochrane risk of bias for RCTs 35 , and a score derived by Murad et al. for case series studies 36 . Pre-specified secondary outcomes were comparison of methodological quality scores of COVID-19 articles by (i) median time to acceptance, (ii) impact factor, (iii) geographical region and (iv) historical comparator. Time of acceptance was defined as the time between submission to acceptance which captures peer review and editorial decisions. Geographical region was stratified into continents including Asia/Oceania, Europe/Africa and Americas (North and South America). Post hoc comparison analysis between COVID-19 and historical control article quality scores were evaluated using Kruskal–Wallis test. Furthermore, good quality of NOS was defined as 3+ on selection and 1+ on comparability, and 2+ on outcome/exposure domains and high-quality case series scores was defined as a score ≥3.5. Due to a small sample size of identified RCTs, they were not included in the comparison analysis.
The finalized dataset was collected on Microsoft Excel v16.44. All statistical analyses were performed using SAS v9.4 (SAS Institute, Inc., Cary, NC, USA). Statistical significance was defined as P < 0.05. All figures were generated using GraphPad Prism v8 (GraphPad Software, La Jolla, CA, USA).
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
The authors can confirm that all relevant data are included in the paper and in Supplementary Data 1 – 2 . The original search was conducted on MEDLINE, Embase and Cochrane Central Register of Controlled Trials.
Chen, Q., Allot, A. & Lu, Z. Keep up with the latest coronavirus research. Nature 579 , 193 (2020).
Article ADS CAS Google Scholar
Mahase, E. Covid-19: 146 researchers raise concerns over chloroquine study that halted WHO trial. BMJ https://doi.org/10.1136/bmj.m2197 (2020).
Chalmers, I. & Glasziou, P. Avoidable waste in the production and reporting of research evidence. Lancet 374 , 86–89 (2009).
Article Google Scholar
Burns, P. B., Rohrich, R. J. & Chung, K. C. The levels of evidence and their role in evidence-based medicine. Plast. Reconstr. Surg. 128 , 305–310 (2011).
Article CAS Google Scholar
Alexander, P. E. et al. COVID-19 coronavirus research has overall low methodological quality thus far: case in point for chloroquine/hydroxychloroquine. J. Clin. Epidemiol. 123 , 120–126 (2020).
Barakat, A. F., Shokr, M., Ibrahim, J., Mandrola, J. & Elgendy, I. Y. Timeline from receipt to online publication of COVID-19 original research articles. Preprint at medRxiv https://doi.org/10.1101/2020.06.22.20137653 (2020).
Chan, A.-W. et al. Increasing value and reducing waste: addressing inaccessible research. Lancet 383 , 257–266 (2014).
Ioannidis, J. P. A. et al. Increasing value and reducing waste in research design, conduct, and analysis. Lancet 383 , 166–175 (2014).
Chalmers, I. et al. How to increase value and reduce waste when research priorities are set. Lancet 383 , 156–165 (2014).
Salman, R. A.-S. et al. Increasing value and reducing waste in biomedical research regulation and management. Lancet 383 , 176–185 (2014).
Glasziou, P. et al. Reducing waste from incomplete or unusable reports of biomedical research. Lancet 383 , 267–276 (2014).
Bauchner, H. The rush to publication: an editorial and scientific mistake. JAMA 318 , 1109–1110 (2017).
He, X. et al. Temporal dynamics in viral shedding and transmissibility of COVID-19. Nat. Med. 26 , 672–675 (2020).
Guaraldi, G. et al. Tocilizumab in patients with severe COVID-19: a retrospective cohort study. Lancet Rheumatol. 2 , e474–e484 (2020).
Duan, K. et al. Effectiveness of convalescent plasma therapy in severe COVID-19 patients. Proc. Natl Acad. Sci. USA 117 , 9490–9496 (2020).
Shen, C. et al. Treatment of 5 critically Ill patients with COVID-19 with convalescent plasma. JAMA 323 , 1582–1589 (2020).
Beigel, J. H. et al. Remdesivir for the treatment of covid-19—final report. N. Engl. J. Med. 383 , 1813–1826 (2020).
Group, R. C. et al. Dexamethasone in hospitalized patients with Covid-19—preliminary report. N. Engl. J. Med. https://doi.org/10.1056/NEJMoa2021436 (2020).
Ramirez, F. D. et al. Methodological rigor in preclinical cardiovascular studies: targets to enhance reproducibility and promote research translation. Circ. Res 120 , 1916–1926 (2017).
Heneghan, C. et al. Evidence based medicine manifesto for better healthcare. BMJ 357 , j2973 (2017).
Mehra, M. R., Desai, S. S., Ruschitzka, F. & Patel, A. N. RETRACTED: hydroxychloroquine or chloroquine with or without a macrolide for treatment of COVID-19: a multinational registry analysis. Lancet https://doi.org/10.1016/S0140-6736(20)31180-6 (2020).
Servick, K. & Enserink, M. The pandemic’s first major research scandal erupts. Science 368 , 1041–1042 (2020).
Mehra, M. R., Desai, S. S., Kuy, S., Henry, T. D. & Patel, A. N. Retraction: Cardiovascular disease, drug therapy, and mortality in Covid-19. N. Engl. J. Med. 382 , 2582–2582, https://doi.org/10.1056/NEJMoa2007621. (2020).
Article PubMed Google Scholar
Boulware, D. R. et al. A randomized trial of hydroxychloroquine as postexposure prophylaxis for Covid-19. N. Engl. J. Med. 383 , 517–525 (2020).
Gautret, P. et al. Clinical and microbiological effect of a combination of hydroxychloroquine and azithromycin in 80 COVID-19 patients with at least a six-day follow up: a pilot observational study. Travel Med. Infect. Dis. 34 , 101663–101663 (2020).
Geleris, J. et al. Observational study of hydroxychloroquine in hospitalized patients with Covid-19. N. Engl. J. Med. 382 , 2411–2418 (2020).
Borba, M. G. S. et al. Effect of high vs low doses of chloroquine diphosphate as adjunctive therapy for patients hospitalized with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection: a randomized clinical trial. JAMA Netw. Open 3 , e208857–e208857 (2020).
Mercuro, N. J. et al. Risk of QT interval prolongation associated with use of hydroxychloroquine with or without concomitant azithromycin among hospitalized patients testing positive for coronavirus disease 2019 (COVID-19). JAMA Cardiol. 5 , 1036–1041 (2020).
Molina, J. M. et al. No evidence of rapid antiviral clearance or clinical benefit with the combination of hydroxychloroquine and azithromycin in patients with severe COVID-19 infection. Médecine et. Maladies Infectieuses 50 , 384 (2020).
Group, R. C. et al. Effect of hydroxychloroquine in hospitalized patients with Covid-19. N. Engl. J. Med . 383, 2030–2040 (2020).
Shors, T. & McFadden, S. H. 1918 influenza: a Winnebago County, Wisconsin perspective. Clin. Med. Res. 7 , 147–156 (2009).
Stolberg, S. A Mad Scramble to Stock Millions of Malaria Pills, Likely for Nothing (The New York Times, 2020).
Hernandez, A. V., Roman, Y. M., Pasupuleti, V., Barboza, J. J. & White, C. M. Hydroxychloroquine or chloroquine for treatment or prophylaxis of COVID-19: a living systematic review. Ann. Int. Med. 173 , 287–296 (2020).
Glasziou, P. & Chalmers, I. Research waste is still a scandal—an essay by Paul Glasziou and Iain Chalmers. BMJ 363 , k4645 (2018).
Higgins, J. P. T. et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343 , d5928 (2011).
Murad, M. H., Sultan, S., Haffar, S. & Bazerbachi, F. Methodological quality and synthesis of case series and case reports. BMJ Evid. Based Med. 23 , 60–63 (2018).
Wells, G. S. B. et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analysis. http://wwwohrica/programs/clinical_epidemiology/oxfordasp (2004).
Whiting, P. F. et al. QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann. Intern. Med. 155 , 529–536 (2011).
Sanderson, S., Tatt, I. D. & Higgins, J. P. Tools for assessing quality and susceptibility to bias in observational studies in epidemiology: a systematic review and annotated bibliography. Int. J. Epidemiol. 36 , 666–676 (2007).
Stang, A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 25 , 603–605 (2010).
Guyatt, G. et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J. Clin. Epidemiol. 64 , 383–394 (2011).
Quigley, J. M., Thompson, J. C., Halfpenny, N. J. & Scott, D. A. Critical appraisal of nonrandomized studies-A review of recommended and commonly used tools. J. Evaluation Clin. Pract. 25 , 44–52 (2019).
Sterne, J. A. et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 355 , i4919 (2016).
Hamajima, N. et al. Case-control studies: matched controls or all available controls? J. Clin. Epidemiol. 47 , 971–975 (1994).
Download references
Acknowledgements
This study received no specific funding or grant from any agency in the public, commercial, or not-for-profit sectors. R.G.J. was supported by the Vanier CIHR Canada Graduate Scholarship. F.D.R. was supported by a CIHR Banting Postdoctoral Fellowship and a Royal College of Physicians and Surgeons of Canada Detweiler Travelling Fellowship. The funder/sponsor(s) had no role in design and conduct of the study, collection, analysis and interpretation of the data.
Author information
These authors contributed equally: Richard G. Jung, Pietro Di Santo.
Authors and Affiliations
CAPITAL Research Group, University of Ottawa Heart Institute, Ottawa, Ontario, Canada
Richard G. Jung, Pietro Di Santo, F. Daniel Ramirez & Trevor Simard
Vascular Biology and Experimental Medicine Laboratory, University of Ottawa Heart Institute, Ottawa, Ontario, Canada
Richard G. Jung, Pietro Di Santo, Trevor Simard & Benjamin Hibbert
Department of Cellular and Molecular Medicine, Faculty of Medicine, University of Ottawa, Ottawa, Ontario, Canada
Richard G. Jung, Trevor Simard & Benjamin Hibbert
Division of Cardiology, University of Ottawa Heart Institute, Ottawa, Ontario, Canada
Pietro Di Santo, Simon Parlow, F. Daniel Ramirez, Trevor Simard & Benjamin Hibbert
School of Epidemiology and Public Health, University of Ottawa, Ottawa, Ontario, Canada
Pietro Di Santo
Faculty of Medicine, University of Ottawa, Ontario, Canada
Cole Clifford & Stephanie Skanes
Department of Medicine, Cumming School of Medicine, Calgary, Alberta, Canada
Graeme Prosperi-Porta
Division of Internal Medicine, The Ottawa Hospital, Ottawa, Ontario, Canada
Berkman Library, University of Ottawa Heart Institute, Ottawa, Ontario, Canada
Sarah Visintini
Hôpital Cardiologique du Haut-Lévêque, CHU Bordeaux, Bordeaux-Pessac, France
F. Daniel Ramirez
L’Institut de Rythmologie et Modélisation Cardiaque (LIRYC), University of Bordeaux, Bordeaux, France
Department of Cardiovascular Medicine, Mayo Clinic, Rochester, MN, USA
Trevor Simard
You can also search for this author in PubMed Google Scholar
Contributions
R.G.J., P.D.S., S.V., F.D.R., T.S. and B.H. participated in the study conception and design. Data acquisition, analysis and interpretation were performed by R.G.J., P.D.S., C.C., G.P.P., S.P., S.S., A.H., F.D.R., T.S. and B.H. Statistical analysis was performed by R.G.J., P.D.S. and B.H. The manuscript was drafted by R.G.J., P.D.S., F.D.R., T.S. and B.H. All authors approved the final version of the manuscript and agree to be accountable to all aspects of the work.
Corresponding author
Correspondence to Benjamin Hibbert .
Ethics declarations
Competing interests.
B.H. reports funding as a clinical trial investigator from Abbott, Boston Scientific and Edwards Lifesciences outside of the submitted work. The remaining authors declare no competing interests.
Additional information
Peer review information Nature Communications Ian White and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information, peer review file, description of additional supplementary files, supplementary data 1, supplementary data 2, reporting summary, rights and permissions.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/ .
Reprints and permissions
About this article
Cite this article.
Jung, R.G., Di Santo, P., Clifford, C. et al. Methodological quality of COVID-19 clinical research. Nat Commun 12 , 943 (2021). https://doi.org/10.1038/s41467-021-21220-5
Download citation
Received : 16 July 2020
Accepted : 13 January 2021
Published : 11 February 2021
DOI : https://doi.org/10.1038/s41467-021-21220-5
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
This article is cited by
Exploring covid-19 research credibility among spanish scientists.
- Eduardo Garcia-Garzon
- Ariadna Angulo-Brunet
- Guido Corradi
Current Psychology (2024)
Primary health care research in COVID-19: analysis of the protocols reviewed by the ethics committee of IDIAPJGol, Catalonia
- Anna Moleras-Serra
- Rosa Morros-Pedros
- Ainhoa Gómez-Lumbreras
BMC Primary Care (2023)
Identifying patterns of reported findings on long-term cardiac complications of COVID-19: a systematic review and meta-analysis
- Chenya Zhao
BMC Medicine (2023)
Implications for implementation and adoption of telehealth in developing countries: a systematic review of China’s practices and experiences
- Jiancheng Ye
- Molly Beestrum
npj Digital Medicine (2023)
Neuropsychological deficits in patients with persistent COVID-19 symptoms: a systematic review and meta-analysis
- Saioa Sobrino-Relaño
- Yolanda Balboa-Bandeira
- Natalia Ojeda
Scientific Reports (2023)
By submitting a comment you agree to abide by our Terms and Community Guidelines . If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies
Sign up for the Nature Briefing newsletter — what matters in science, free to your inbox daily.
Coronavirus disease 2019 (COVID-19): A literature review
Affiliations.
- 1 Medical Research Unit, School of Medicine, Universitas Syiah Kuala, Banda Aceh, Indonesia; Tropical Disease Centre, School of Medicine, Universitas Syiah Kuala, Banda Aceh, Indonesia; Department of Microbiology, School of Medicine, Universitas Syiah Kuala, Banda Aceh, Indonesia. Electronic address: [email protected].
- 2 Division of Infectious Diseases, AichiCancer Center Hospital, Chikusa-ku Nagoya, Japan. Electronic address: [email protected].
- 3 Department of Family Medicine, School of Medicine, Universitas Syiah Kuala, Banda Aceh, Indonesia. Electronic address: [email protected].
- 4 Department of Pulmonology and Respiratory Medicine, School of Medicine, Universitas Syiah Kuala, Banda Aceh, Indonesia. Electronic address: [email protected].
- 5 School of Medicine, The University of Western Australia, Perth, Australia. Electronic address: [email protected].
- 6 Siem Reap Provincial Health Department, Ministry of Health, Siem Reap, Cambodia. Electronic address: [email protected].
- 7 Department of Microbiology and Parasitology, Faculty of Medicine and Health Sciences, Warmadewa University, Denpasar, Indonesia; Department of Medical Microbiology and Immunology, University of California, Davis, CA, USA. Electronic address: [email protected].
- 8 Medical Research Unit, School of Medicine, Universitas Syiah Kuala, Banda Aceh, Indonesia; Tropical Disease Centre, School of Medicine, Universitas Syiah Kuala, Banda Aceh, Indonesia; Department of Microbiology, School of Medicine, Universitas Syiah Kuala, Banda Aceh, Indonesia; Department of Clinical Microbiology, School of Medicine, Universitas Syiah Kuala, Banda Aceh, Indonesia. Electronic address: [email protected].
- 9 Department of Epidemiology, University of Michigan, Ann Arbor, Michigan, MI 48109, USA. Electronic address: [email protected].
- 10 Medical Research Unit, School of Medicine, Universitas Syiah Kuala, Banda Aceh, Indonesia; Tropical Disease Centre, School of Medicine, Universitas Syiah Kuala, Banda Aceh, Indonesia; Department of Microbiology, School of Medicine, Universitas Syiah Kuala, Banda Aceh, Indonesia. Electronic address: [email protected].
- PMID: 32340833
- PMCID: PMC7142680
- DOI: 10.1016/j.jiph.2020.03.019
In early December 2019, an outbreak of coronavirus disease 2019 (COVID-19), caused by a novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), occurred in Wuhan City, Hubei Province, China. On January 30, 2020 the World Health Organization declared the outbreak as a Public Health Emergency of International Concern. As of February 14, 2020, 49,053 laboratory-confirmed and 1,381 deaths have been reported globally. Perceived risk of acquiring disease has led many governments to institute a variety of control measures. We conducted a literature review of publicly available information to summarize knowledge about the pathogen and the current epidemic. In this literature review, the causative agent, pathogenesis and immune responses, epidemiology, diagnosis, treatment and management of the disease, control and preventions strategies are all reviewed.
Keywords: 2019-nCoV; COVID-19; Novel coronavirus; Outbreak; SARS-CoV-2.
Copyright © 2020 The Authors. Published by Elsevier Ltd.. All rights reserved.
Publication types
- Betacoronavirus
- Clinical Trials as Topic
- Coronavirus Infections* / epidemiology
- Coronavirus Infections* / immunology
- Coronavirus Infections* / therapy
- Coronavirus Infections* / virology
- Disease Outbreaks* / prevention & control
- Pneumonia, Viral* / epidemiology
- Pneumonia, Viral* / immunology
- Pneumonia, Viral* / therapy
- Pneumonia, Viral* / virology
- Introduction
- Observations
- Clinical Presentation
- Assessment and Diagnosis
- Disparities
- Prevention and Vaccine Development
- Conclusions
- Article Information
Based on data from World Health Organization (WHO) COVID-19 situation reports. The COVID-19 outbreak was first recognized in Wuhan, China, in early December 2019. The number of confirmed COVID-19 cases is displayed by date of report and WHO region. SARS-CoV-2 indicates severe acute respiratory syndrome coronavirus 2.
Current understanding of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)–induced host immune response. SARS-CoV-2 targets cells through the viral structural spike (S) protein that binds to the angiotensin-converting enzyme 2 (ACE2) receptor. The serine protease type 2 transmembrane serine proteas (TMPRSS2) in the host cell further promotes viral uptake by cleaving ACE2 and activating the SARS-CoV-2 S protein. In the early stage, viral copy numbers can be high in the lower respiratory tract. Inflammatory signaling molecules are released by infected cells and alveolar macrophages in addition to recruited T lymphocytes, monocytes, and neutrophils. In the late stage, pulmonary edema can fill the alveolar spaces with hyaline membrane formation, compatible with early-phase acute respiratory distress syndrome.
A, Transverse thin-section computed tomographic scan of a 76-year-old man, 5 days after symptom onset, showing subpleural ground-glass opacity and consolidation with subpleural sparing. B, Transverse thin-section computed tomographic scan of a 76-year-old man, 21 days after symptom onset, showing bilateral and peripheral predominant consolidation, ground-glass with reticulation, and bronchodilatation. C, Pathological manifestations of lung tissue in a patient with severe pneumonia caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) showing interstitial mononuclear inflammatory infiltrates dominated by lymphocytes (magnification, ×10). D, Pathological manifestations of lung tissue in a patient with severe pneumonia caused by SARS-CoV-2 showing diffuse alveolar damage with edema and fibrine deposition, indicating acute respiratory distress syndrome with early fibrosis (magnification, ×10). Images courtesy of Inge A. H. van den Berk, MD (Department of Radiology, Amsterdam UMC), and Bernadette Schurink, MD (Department of Pathology, Amsterdam UMC).
Eric Topol, MD, Scripps Research EVP and omnivorous science health care and tech commentator, discusses the evolving COVID-19 pandemic. Recorded July 23, 2020.
- Pharmacologic Treatments for Coronavirus Disease 2019 (COVID-19) JAMA Review May 12, 2020 This narrative review summarizes what is currently known about how SARS-CoV-2 infects cells and causes disease as a basis for considering whether chloroquine, remdisivir and other antivirals, or other existing drugs might be effective treatment for coronavirus disease 2019 (COVID-19). James M. Sanders, PhD, PharmD; Marguerite L. Monogue, PharmD; Tomasz Z. Jodlowski, PharmD; James B. Cutrell, MD
- Ensuring Scientific Integrity and Public Confidence in the Search for Effective COVID-19 Treatment JAMA Viewpoint May 19, 2020 This Viewpoint discusses the risks to patients and public health posed by the FDA’s politically pressured Emergency Use Authorization (EUA) of chloroquine and hydroxychloroquine for COVID-19 treatment, and proposes principles to follow to ensure new therapies are studied properly and quickly to maximize benefits and minimize risks to patients. Jesse L. Goodman, MD, MPH; Luciana Borio, MD
- Monoclonal Antibodies for Prevention and Treatment of COVID-19 JAMA Viewpoint July 14, 2020 This Viewpoint discusses the potential role of neutralizing monoclonal antibodies (MAbs) as a treatment for coronavirus disease 2019 (COVID-19) and as a means of prevention in high-risk populations, and it also raises possible limitations of the approach that need to be disproven or addressed for the strategy to be effective. Mary Marovich, MD; John R. Mascola, MD; Myron S. Cohen, MD
- Presence of Genetic Variants Among Young Men With Severe COVID-19 JAMA Preliminary Communication August 18, 2020 This case series describes rare putative X-chromosomal loss-of-function variants associated with impaired peripheral mononuclear blood cell interferon signaling in 4 young male patients hospitalized with severe coronavirus disease 2019 (COVID-19) in the Netherlands. Caspar I. van der Made, MD; Annet Simons, PhD; Janneke Schuurs-Hoeijmakers, MD, PhD; Guus van den Heuvel, MD; Tuomo Mantere, PhD; Simone Kersten, MSc; Rosanne C. van Deuren, MSc; Marloes Steehouwer, BSc; Simon V. van Reijmersdal, BSc; Martin Jaeger, PhD; Tom Hofste, BSc; Galuh Astuti, PhD; Jordi Corominas Galbany, PhD; Vyne van der Schoot, MD, PhD; Hans van der Hoeven, MD, PhD; Wanda Hagmolen of ten Have, MD, PhD; Eva Klijn, MD, PhD; Catrien van den Meer, MD; Jeroen Fiddelaers, MD; Quirijn de Mast, MD, PhD; Chantal P. Bleeker-Rovers, MD, PhD; Leo A. B. Joosten, PhD; Helger G. Yntema, PhD; Christian Gilissen, PhD; Marcel Nelen, PhD; Jos W. M. van der Meer, MD, PhD; Han G. Brunner, MD, PhD; Mihai G. Netea, MD, PhD; Frank L. van de Veerdonk, MD, PhD; Alexander Hoischen, PhD
- Strongyloides Hyperinfection Risk in COVID-19 Patients Treated With Dexamethasone JAMA Viewpoint August 18, 2020 Anticipating widespread global use of dexamethasone for COVID-19 in the wake of RECOVERY trial findings, this Viewpoint summarizes the theoretical risk of triggering Strongyloides hyperinfection/dissemination syndrome in people with asymptomatic strongyloidiasis, and proposes an algorithm to for assessing and managing the risk in outpatient and hospital settings. William M. Stauffer, MD, MSPH; Jonathan D. Alpern, MD; Patricia F. Walker, MD, DTM&H
- Patient Information: COVID-19 JAMA JAMA Patient Page August 25, 2020 This JAMA Patient Page provides an overview of COVID-19 transmission, symptoms, diagnosis, disease course, and treatment. W. Joost Wiersinga, MD, PhD, MBA; Hallie C. Prescott, MD, MSc
- Cytokine Levels in Critically Ill Patients With COVID-19 and Other Conditions JAMA Research Letter October 20, 2020 This study compares levels of tumor necrosis factor α, IL-6, and IL-8 in critically ill patients with coronavirus disease 2019 (COVID-19) vs those with other critical illness to better characterize the contribution of cytokine storm to COVID-19 pathophysiology. Matthijs Kox, PhD; Nicole J. B. Waalders, BSc; Emma J. Kooistra, BSc; Jelle Gerretsen, BSc; Peter Pickkers, MD, PhD
- Racial/Ethnic Variation in Nasal Transmembrane Serine Protease 2 ( TMPRSS2 ) Gene Expression Facilitating Coronavirus Entry JAMA Research Letter October 20, 2020 This cross-sectional study used nasal epithelium collected in 2015-2018 to compare expression of TMPRSS2, a facilitator of SARS-CoV-2 viral entry and spread, among Asian, Black, Latino, and White patients as well as patients of mixed race/ethnicity within a New York City health system. Supinda Bunyavanich, MD, MPH, MPhil; Chantal Grant, MD; Alfin Vicencio, MD
- Therapy for Early COVID-19—A Critical Need JAMA Viewpoint December 1, 2020 In this Viewpoint, Fauci and NIAID colleagues review leading candidates for treatment of mild to moderate coronavirus disease 2019 (COVID-19) to prevent disease progression and longer-term complications, including emerging antiviral drugs, immune-modulating agents, and antibody-based therapies, and the challenges of developing randomized trials to rapidly evaluate the safety and efficacy of each. Peter S. Kim, MD; Sarah W. Read, MD, MHS; Anthony S. Fauci, MD
See More About
Select your interests.
Customize your JAMA Network experience by selecting one or more topics from the list below.
- Academic Medicine
- Acid Base, Electrolytes, Fluids
- Allergy and Clinical Immunology
- American Indian or Alaska Natives
- Anesthesiology
- Anticoagulation
- Art and Images in Psychiatry
- Artificial Intelligence
- Assisted Reproduction
- Bleeding and Transfusion
- Caring for the Critically Ill Patient
- Challenges in Clinical Electrocardiography
- Climate and Health
- Climate Change
- Clinical Challenge
- Clinical Decision Support
- Clinical Implications of Basic Neuroscience
- Clinical Pharmacy and Pharmacology
- Complementary and Alternative Medicine
- Consensus Statements
- Coronavirus (COVID-19)
- Critical Care Medicine
- Cultural Competency
- Dental Medicine
- Dermatology
- Diabetes and Endocrinology
- Diagnostic Test Interpretation
- Drug Development
- Electronic Health Records
- Emergency Medicine
- End of Life, Hospice, Palliative Care
- Environmental Health
- Equity, Diversity, and Inclusion
- Facial Plastic Surgery
- Gastroenterology and Hepatology
- Genetics and Genomics
- Genomics and Precision Health
- Global Health
- Guide to Statistics and Methods
- Hair Disorders
- Health Care Delivery Models
- Health Care Economics, Insurance, Payment
- Health Care Quality
- Health Care Reform
- Health Care Safety
- Health Care Workforce
- Health Disparities
- Health Inequities
- Health Policy
- Health Systems Science
- History of Medicine
- Hypertension
- Images in Neurology
- Implementation Science
- Infectious Diseases
- Innovations in Health Care Delivery
- JAMA Infographic
- Law and Medicine
- Leading Change
- Less is More
- LGBTQIA Medicine
- Lifestyle Behaviors
- Medical Coding
- Medical Devices and Equipment
- Medical Education
- Medical Education and Training
- Medical Journals and Publishing
- Mobile Health and Telemedicine
- Narrative Medicine
- Neuroscience and Psychiatry
- Notable Notes
- Nutrition, Obesity, Exercise
- Obstetrics and Gynecology
- Occupational Health
- Ophthalmology
- Orthopedics
- Otolaryngology
- Pain Medicine
- Palliative Care
- Pathology and Laboratory Medicine
- Patient Care
- Patient Information
- Performance Improvement
- Performance Measures
- Perioperative Care and Consultation
- Pharmacoeconomics
- Pharmacoepidemiology
- Pharmacogenetics
- Pharmacy and Clinical Pharmacology
- Physical Medicine and Rehabilitation
- Physical Therapy
- Physician Leadership
- Population Health
- Primary Care
- Professional Well-being
- Professionalism
- Psychiatry and Behavioral Health
- Public Health
- Pulmonary Medicine
- Regulatory Agencies
- Reproductive Health
- Research, Methods, Statistics
- Resuscitation
- Rheumatology
- Risk Management
- Scientific Discovery and the Future of Medicine
- Shared Decision Making and Communication
- Sleep Medicine
- Sports Medicine
- Stem Cell Transplantation
- Substance Use and Addiction Medicine
- Surgical Innovation
- Surgical Pearls
- Teachable Moment
- Technology and Finance
- The Art of JAMA
- The Arts and Medicine
- The Rational Clinical Examination
- Tobacco and e-Cigarettes
- Translational Medicine
- Trauma and Injury
- Treatment Adherence
- Ultrasonography
- Users' Guide to the Medical Literature
- Vaccination
- Venous Thromboembolism
- Veterans Health
- Women's Health
- Workflow and Process
- Wound Care, Infection, Healing
Others Also Liked
- Download PDF
- X Facebook More LinkedIn
Wiersinga WJ , Rhodes A , Cheng AC , Peacock SJ , Prescott HC. Pathophysiology, Transmission, Diagnosis, and Treatment of Coronavirus Disease 2019 (COVID-19) : A Review . JAMA. 2020;324(8):782–793. doi:10.1001/jama.2020.12839
Manage citations:
© 2024
- Permissions
Pathophysiology, Transmission, Diagnosis, and Treatment of Coronavirus Disease 2019 (COVID-19) : A Review
- 1 Division of Infectious Diseases, Department of Medicine, Amsterdam UMC, location AMC, University of Amsterdam, Amsterdam, the Netherlands
- 2 Center for Experimental and Molecular Medicine (CEMM), Amsterdam UMC, location AMC, University of Amsterdam, Amsterdam, the Netherlands
- 3 Department of Intensive Care Medicine, St George's University Hospitals Foundation Trust, London, United Kingdom
- 4 Infection Prevention and Healthcare Epidemiology Unit, Alfred Health, Melbourne, Australia
- 5 School of Public Health and Preventive Medicine, Monash University, Monash University, Melbourne, Australia
- 6 National Infection Service, Public Health England, London, United Kingdom
- 7 Department of Medicine, University of Cambridge, Addenbrooke's Hospital, Cambridge, United Kingdom
- 8 Division of Pulmonary and Critical Care Medicine, University of Michigan, Ann Arbor
- 9 VA Center for Clinical Management Research, Ann Arbor, Michigan
- Review Pharmacologic Treatments for Coronavirus Disease 2019 (COVID-19) James M. Sanders, PhD, PharmD; Marguerite L. Monogue, PharmD; Tomasz Z. Jodlowski, PharmD; James B. Cutrell, MD JAMA
- Viewpoint Ensuring Scientific Integrity and Public Confidence in the Search for Effective COVID-19 Treatment Jesse L. Goodman, MD, MPH; Luciana Borio, MD JAMA
- Viewpoint Monoclonal Antibodies for Prevention and Treatment of COVID-19 Mary Marovich, MD; John R. Mascola, MD; Myron S. Cohen, MD JAMA
- Preliminary Communication Presence of Genetic Variants Among Young Men With Severe COVID-19 Caspar I. van der Made, MD; Annet Simons, PhD; Janneke Schuurs-Hoeijmakers, MD, PhD; Guus van den Heuvel, MD; Tuomo Mantere, PhD; Simone Kersten, MSc; Rosanne C. van Deuren, MSc; Marloes Steehouwer, BSc; Simon V. van Reijmersdal, BSc; Martin Jaeger, PhD; Tom Hofste, BSc; Galuh Astuti, PhD; Jordi Corominas Galbany, PhD; Vyne van der Schoot, MD, PhD; Hans van der Hoeven, MD, PhD; Wanda Hagmolen of ten Have, MD, PhD; Eva Klijn, MD, PhD; Catrien van den Meer, MD; Jeroen Fiddelaers, MD; Quirijn de Mast, MD, PhD; Chantal P. Bleeker-Rovers, MD, PhD; Leo A. B. Joosten, PhD; Helger G. Yntema, PhD; Christian Gilissen, PhD; Marcel Nelen, PhD; Jos W. M. van der Meer, MD, PhD; Han G. Brunner, MD, PhD; Mihai G. Netea, MD, PhD; Frank L. van de Veerdonk, MD, PhD; Alexander Hoischen, PhD JAMA
- Viewpoint Strongyloides Hyperinfection Risk in COVID-19 Patients Treated With Dexamethasone William M. Stauffer, MD, MSPH; Jonathan D. Alpern, MD; Patricia F. Walker, MD, DTM&H JAMA
- JAMA Patient Page Patient Information: COVID-19 W. Joost Wiersinga, MD, PhD, MBA; Hallie C. Prescott, MD, MSc JAMA
- Research Letter Cytokine Levels in Critically Ill Patients With COVID-19 and Other Conditions Matthijs Kox, PhD; Nicole J. B. Waalders, BSc; Emma J. Kooistra, BSc; Jelle Gerretsen, BSc; Peter Pickkers, MD, PhD JAMA
- Research Letter Racial/Ethnic Variation in Nasal Transmembrane Serine Protease 2 ( TMPRSS2 ) Gene Expression Facilitating Coronavirus Entry Supinda Bunyavanich, MD, MPH, MPhil; Chantal Grant, MD; Alfin Vicencio, MD JAMA
- Viewpoint Therapy for Early COVID-19—A Critical Need Peter S. Kim, MD; Sarah W. Read, MD, MHS; Anthony S. Fauci, MD JAMA
Importance The coronavirus disease 2019 (COVID-19) pandemic, due to the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has caused a worldwide sudden and substantial increase in hospitalizations for pneumonia with multiorgan disease. This review discusses current evidence regarding the pathophysiology, transmission, diagnosis, and management of COVID-19.
Observations SARS-CoV-2 is spread primarily via respiratory droplets during close face-to-face contact. Infection can be spread by asymptomatic, presymptomatic, and symptomatic carriers. The average time from exposure to symptom onset is 5 days, and 97.5% of people who develop symptoms do so within 11.5 days. The most common symptoms are fever, dry cough, and shortness of breath. Radiographic and laboratory abnormalities, such as lymphopenia and elevated lactate dehydrogenase, are common, but nonspecific. Diagnosis is made by detection of SARS-CoV-2 via reverse transcription polymerase chain reaction testing, although false-negative test results may occur in up to 20% to 67% of patients; however, this is dependent on the quality and timing of testing. Manifestations of COVID-19 include asymptomatic carriers and fulminant disease characterized by sepsis and acute respiratory failure. Approximately 5% of patients with COVID-19, and 20% of those hospitalized, experience severe symptoms necessitating intensive care. More than 75% of patients hospitalized with COVID-19 require supplemental oxygen. Treatment for individuals with COVID-19 includes best practices for supportive management of acute hypoxic respiratory failure. Emerging data indicate that dexamethasone therapy reduces 28-day mortality in patients requiring supplemental oxygen compared with usual care (21.6% vs 24.6%; age-adjusted rate ratio, 0.83 [95% CI, 0.74-0.92]) and that remdesivir improves time to recovery (hospital discharge or no supplemental oxygen requirement) from 15 to 11 days. In a randomized trial of 103 patients with COVID-19, convalescent plasma did not shorten time to recovery. Ongoing trials are testing antiviral therapies, immune modulators, and anticoagulants. The case-fatality rate for COVID-19 varies markedly by age, ranging from 0.3 deaths per 1000 cases among patients aged 5 to 17 years to 304.9 deaths per 1000 cases among patients aged 85 years or older in the US. Among patients hospitalized in the intensive care unit, the case fatality is up to 40%. At least 120 SARS-CoV-2 vaccines are under development. Until an effective vaccine is available, the primary methods to reduce spread are face masks, social distancing, and contact tracing. Monoclonal antibodies and hyperimmune globulin may provide additional preventive strategies.
Conclusions and Relevance As of July 1, 2020, more than 10 million people worldwide had been infected with SARS-CoV-2. Many aspects of transmission, infection, and treatment remain unclear. Advances in prevention and effective management of COVID-19 will require basic and clinical investigation and public health and clinical interventions.
The coronavirus disease 2019 (COVID-19) pandemic has caused a sudden significant increase in hospitalizations for pneumonia with multiorgan disease. COVID-19 is caused by the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). SARS-CoV-2 infection may be asymptomatic or it may cause a wide spectrum of symptoms, such as mild symptoms of upper respiratory tract infection and life-threatening sepsis. COVID-19 first emerged in December 2019, when a cluster of patients with pneumonia of unknown cause was recognized in Wuhan, China. As of July 1, 2020, SARS-CoV-2 has affected more than 200 countries, resulting in more than 10 million identified cases with 508 000 confirmed deaths ( Figure 1 ). This review summarizes current evidence regarding pathophysiology, transmission, diagnosis, and management of COVID-19.
We searched PubMed, LitCovid, and MedRxiv using the search terms coronavirus , severe acute respiratory syndrome coronavirus 2 , 2019-nCoV , SARS-CoV-2 , SARS-CoV , MERS-CoV , and COVID-19 for studies published from January 1, 2002, to June 15, 2020, and manually searched the references of select articles for additional relevant articles. Ongoing or completed clinical trials were identified using the disease search term coronavirus infection on ClinicalTrials.gov, the Chinese Clinical Trial Registry, and the International Clinical Trials Registry Platform. We selected articles relevant to a general medicine readership, prioritizing randomized clinical trials, systematic reviews, and clinical practice guidelines.
Coronaviruses are large, enveloped, single-stranded RNA viruses found in humans and other mammals, such as dogs, cats, chicken, cattle, pigs, and birds. Coronaviruses cause respiratory, gastrointestinal, and neurological disease. The most common coronaviruses in clinical practice are 229E, OC43, NL63, and HKU1, which typically cause common cold symptoms in immunocompetent individuals. SARS-CoV-2 is the third coronavirus that has caused severe disease in humans to spread globally in the past 2 decades. 1 The first coronavirus that caused severe disease was severe acute respiratory syndrome (SARS), which was thought to originate in Foshan, China, and resulted in the 2002-2003 SARS-CoV pandemic. 2 The second was the coronavirus-caused Middle East respiratory syndrome (MERS), which originated from the Arabian peninsula in 2012. 3
SARS-CoV-2 has a diameter of 60 nm to 140 nm and distinctive spikes, ranging from 9 nm to 12 nm, giving the virions the appearance of a solar corona ( Figure 2 ). 4 Through genetic recombination and variation, coronaviruses can adapt to and infect new hosts. Bats are thought to be a natural reservoir for SARS-CoV-2, but it has been suggested that humans became infected with SARS-CoV-2 via an intermediate host, such as the pangolin. 5 , 6
Early in infection, SARS-CoV-2 targets cells, such as nasal and bronchial epithelial cells and pneumocytes, through the viral structural spike (S) protein that binds to the angiotensin-converting enzyme 2 (ACE2) receptor 7 ( Figure 2 ). The type 2 transmembrane serine protease (TMPRSS2), present in the host cell, promotes viral uptake by cleaving ACE2 and activating the SARS-CoV-2 S protein, which mediates coronavirus entry into host cells. 7 ACE2 and TMPRSS2 are expressed in host target cells, particularly alveolar epithelial type II cells. 8 , 9 Similar to other respiratory viral diseases, such as influenza, profound lymphopenia may occur in individuals with COVID-19 when SARS-CoV-2 infects and kills T lymphocyte cells. In addition, the viral inflammatory response, consisting of both the innate and the adaptive immune response (comprising humoral and cell-mediated immunity), impairs lymphopoiesis and increases lymphocyte apoptosis. Although upregulation of ACE2 receptors from ACE inhibitor and angiotensin receptor blocker medications has been hypothesized to increase susceptibility to SARS-CoV-2 infection, large observational cohorts have not found an association between these medications and risk of infection or hospital mortality due to COVID-19. 10 , 11 For example, in a study 4480 patients with COVID-19 from Denmark, previous treatment with ACE inhibitors or angiotensin receptor blockers was not associated with mortality. 11
In later stages of infection, when viral replication accelerates, epithelial-endothelial barrier integrity is compromised. In addition to epithelial cells, SARS-CoV-2 infects pulmonary capillary endothelial cells, accentuating the inflammatory response and triggering an influx of monocytes and neutrophils. Autopsy studies have shown diffuse thickening of the alveolar wall with mononuclear cells and macrophages infiltrating airspaces in addition to endothelialitis. 12 Interstitial mononuclear inflammatory infiltrates and edema develop and appear as ground-glass opacities on computed tomographic imaging. Pulmonary edema filling the alveolar spaces with hyaline membrane formation follows, compatible with early-phase acute respiratory distress syndrome (ARDS). 12 Bradykinin-dependent lung angioedema may contribute to disease. 13 Collectively, endothelial barrier disruption, dysfunctional alveolar-capillary oxygen transmission, and impaired oxygen diffusion capacity are characteristic features of COVID-19.
In severe COVID-19, fulminant activation of coagulation and consumption of clotting factors occur. 14 , 15 A report from Wuhan, China, indicated that 71% of 183 individuals who died of COVID-19 met criteria for diffuse intravascular coagulation. 14 Inflamed lung tissues and pulmonary endothelial cells may result in microthrombi formation and contribute to the high incidence of thrombotic complications, such as deep venous thrombosis, pulmonary embolism, and thrombotic arterial complications (eg, limb ischemia, ischemic stroke, myocardial infarction) in critically ill patients. 16 The development of viral sepsis, defined as life-threatening organ dysfunction caused by a dysregulated host response to infection, may further contribute to multiorgan failure.
Epidemiologic data suggest that droplets expelled during face-to-face exposure during talking, coughing, or sneezing is the most common mode of transmission ( Box 1 ). Prolonged exposure to an infected person (being within 6 feet for at least 15 minutes) and briefer exposures to individuals who are symptomatic (eg, coughing) are associated with higher risk for transmission, while brief exposures to asymptomatic contacts are less likely to result in transmission. 25 Contact surface spread (touching a surface with virus on it) is another possible mode of transmission. Transmission may also occur via aerosols (smaller droplets that remain suspended in air), but it is unclear if this is a significant source of infection in humans outside of a laboratory setting. 26 , 27 The existence of aerosols in physiological states (eg, coughing) or the detection of nucleic acid in the air does not mean that small airborne particles are infectious. 28 Maternal COVID-19 is currently believed to be associated with low risk for vertical transmission. In most reported series, the mothers' infection with SARS-CoV-2 occurred in the third trimester of pregnancy, with no maternal deaths and a favorable clinical course in the neonates. 29 - 31
Transmission, Symptoms, and Complications of Coronavirus Disease 2019 (COVID-19)
Transmission 17 of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) occurs primarily via respiratory droplets from face-to-face contact and, to a lesser degree, via contaminated surfaces. Aerosol spread may occur, but the role of aerosol spread in humans remains unclear. An estimated 48% to 62% of transmission may occur via presymptomatic carriers.
Common symptoms 18 in hospitalized patients include fever (70%-90%), dry cough (60%-86%), shortness of breath (53%-80%), fatigue (38%), myalgias (15%-44%), nausea/vomiting or diarrhea (15%-39%), headache, weakness (25%), and rhinorrhea (7%). Anosmia or ageusia may be the sole presenting symptom in approximately 3% of individuals with COVID-19.
Common laboratory abnormalities 19 among hospitalized patients include lymphopenia (83%), elevated inflammatory markers (eg, erythrocyte sedimentation rate, C-reactive protein, ferritin, tumor necrosis factor-α, IL-1, IL-6), and abnormal coagulation parameters (eg, prolonged prothrombin time, thrombocytopenia, elevated D-dimer [46% of patients], low fibrinogen).
Common radiographic findings of individuals with COVID-19 include bilateral, lower-lobe predominate infiltrates on chest radiographic imaging and bilateral, peripheral, lower-lobe ground-glass opacities and/or consolidation on chest computed tomographic imaging.
Common complications 18 , 20 - 24 among hospitalized patients with COVID-19 include pneumonia (75%); acute respiratory distress syndrome (15%); acute liver injury, characterized by elevations in aspartate transaminase, alanine transaminase, and bilirubin (19%); cardiac injury, including troponin elevation (7%-17%), acute heart failure, dysrhythmias, and myocarditis; prothrombotic coagulopathy resulting in venous and arterial thromboembolic events (10%-25%); acute kidney injury (9%); neurologic manifestations, including impaired consciousness (8%) and acute cerebrovascular disease (3%); and shock (6%).
Rare complications among critically ill patients with COVID-19 include cytokine storm and macrophage activation syndrome (ie, secondary hemophagocytic lymphohistiocytosis).
The clinical significance of SARS-CoV-2 transmission from inanimate surfaces is difficult to interpret without knowing the minimum dose of virus particles that can initiate infection. Viral load appears to persist at higher levels on impermeable surfaces, such as stainless steel and plastic, than permeable surfaces, such as cardboard. 32 Virus has been identified on impermeable surfaces for up to 3 to 4 days after inoculation. 32 Widespread viral contamination of hospital rooms has been documented. 28 However, it is thought that the amount of virus detected on surfaces decays rapidly within 48 to 72 hours. 32 Although the detection of virus on surfaces reinforces the potential for transmission via fomites (objects such as a doorknob, cutlery, or clothing that may be contaminated with SARS-CoV-2) and the need for adequate environmental hygiene, droplet spread via face-to-face contact remains the primary mode of transmission.
Viral load in the upper respiratory tract appears to peak around the time of symptom onset and viral shedding begins approximately 2 to 3 days prior to the onset of symptoms. 33 Asymptomatic and presymptomatic carriers can transmit SARS-CoV-2. 34 , 35 In Singapore, presymptomatic transmission has been described in clusters of patients with close contact (eg, through churchgoing or singing class) approximately 1 to 3 days before the source patient developed symptoms. 34 Presymptomatic transmission is thought to be a major contributor to the spread of SARS-CoV-2. Modeling studies from China and Singapore estimated the percentage of infections transmitted from a presymptomatic individual as 48% to 62%. 17 Pharyngeal shedding is high during the first week of infection at a time in which symptoms are still mild, which might explain the efficient transmission of SARS-CoV-2, because infected individuals can be infectious before they realize they are ill. 36 Although studies have described rates of asymptomatic infection, ranging from 4% to 32%, it is unclear whether these reports represent truly asymptomatic infection by individuals who never develop symptoms, transmission by individuals with very mild symptoms, or transmission by individuals who are asymptomatic at the time of transmission but subsequently develop symptoms. 37 - 39 A systematic review on this topic suggested that true asymptomatic infection is probably uncommon. 38
Although viral nucleic acid can be detectable in throat swabs for up to 6 weeks after the onset of illness, several studies suggest that viral cultures are generally negative for SARS-CoV-2 8 days after symptom onset. 33 , 36 , 40 This is supported by epidemiological studies that have shown that transmission did not occur to contacts whose exposure to the index case started more than 5 days after the onset of symptoms in the index case. 41 This suggests that individuals can be released from isolation based on clinical improvement. The Centers for Disease Control and Prevention recommend isolating for at least 10 days after symptom onset and 3 days after improvement of symptoms. 42 However, there remains uncertainty about whether serial testing is required for specific subgroups, such as immunosuppressed patients or critically ill patients for whom symptom resolution may be delayed or older adults residing in short- or long-term care facilities.
The mean (interquartile range) incubation period (the time from exposure to symptom onset) for COVID-19 is approximately 5 (2-7) days. 43 , 44 Approximately 97.5% of individuals who develop symptoms will do so within 11.5 days of infection. 43 The median (interquartile range) interval from symptom onset to hospital admission is 7 (3-9) days. 45 The median age of hospitalized patients varies between 47 and 73 years, with most cohorts having a male preponderance of approximately 60%. 44 , 46 , 47 Among patients hospitalized with COVID-19, 74% to 86% are aged at least 50 years. 45 , 47
COVID-19 has various clinical manifestations ( Box 1 and Box 2 ). In a study of 44 672 patients with COVID-19 in China, 81% of patients had mild manifestations, 14% had severe manifestations, and 5% had critical manifestations (defined by respiratory failure, septic shock, and/or multiple organ dysfunction). 48 A study of 20 133 individuals hospitalized with COVID-19 in the UK reported that 17.1% were admitted to high-dependency or intensive care units (ICUs). 47
Commonly Asked Questions About Coronavirus Disease 2019 (COVID-19)
How is severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) most commonly transmitted?
SARS-CoV-2 is most commonly spread via respiratory droplets (eg, from coughing, sneezing, shouting) during face-to-face exposure or by surface contamination.
What are the most common symptoms of COVID-19?
The 3 most common symptoms are fever, cough, and shortness of breath. Additional symptoms include weakness, fatigue, nausea, vomiting, diarrhea, changes to taste and smell.
How is the diagnosis made?
Diagnosis of COVID-19 is typically made by polymerase chain reaction testing of a nasopharyngeal swab. However, given the possibility of false-negative test results, clinical, laboratory, and imaging findings may also be used to make a presumptive diagnosis for individuals for whom there is a high index of clinical suspicion of infection.
What are current evidence-based treatments for individuals with COVID-19?
Supportive care, including supplemental oxygen, is the main treatment for most patients. Recent trials indicate that dexamethasone decreases mortality (subgroup analysis suggests benefit is limited to patients who require supplemental oxygen and who have symptoms for >7 d) and remdesivir improves time to recovery (subgroup analysis suggests benefit is limited to patients not receiving mechanical ventilation).
What percentage of people are asymptomatic carriers, and how important are they in transmitting the disease?
True asymptomatic infection is believed to be uncommon. The average time from exposure to symptoms onset is 5 days, and up to 62% of transmission may occur prior to the onset of symptoms.
Are masks effective at preventing spread?
Yes. Face masks reduce the spread of viral respiratory infection. N95 respirators and surgical masks both provide substantial protection (compared with no mask), and surgical masks provide greater protection than cloth masks. However, physical distancing is also associated with substantial reduction of viral transmission, with greater distances providing greater protection. Additional measures such as hand and environmental disinfection are also important.
Although only approximately 25% of infected patients have comorbidities, 60% to 90% of hospitalized infected patients have comorbidities. 45 - 49 The most common comorbidities in hospitalized patients include hypertension (present in 48%-57% of patients), diabetes (17%-34%), cardiovascular disease (21%-28%), chronic pulmonary disease (4%-10%), chronic kidney disease (3%-13%), malignancy (6%-8%), and chronic liver disease (<5%). 45 , 46 , 49
The most common symptoms in hospitalized patients are fever (up to 90% of patients), dry cough (60%-86%), shortness of breath (53%-80%), fatigue (38%), nausea/vomiting or diarrhea (15%-39%), and myalgia (15%-44%). 18 , 44 - 47 , 49 , 50 Patients can also present with nonclassical symptoms, such as isolated gastrointestinal symptoms. 18 Olfactory and/or gustatory dysfunctions have been reported in 64% to 80% of patients. 51 - 53 Anosmia or ageusia may be the sole presenting symptom in approximately 3% of patients. 53
Complications of COVID-19 include impaired function of the heart, brain, lung, liver, kidney, and coagulation system. COVID-19 can lead to myocarditis, cardiomyopathy, ventricular arrhythmias, and hemodynamic instability. 20 , 54 Acute cerebrovascular disease and encephalitis are observed with severe illness (in up to 8% of patients). 21 , 52 Venous and arterial thromboembolic events occur in 10% to 25% in hospitalized patients with COVID-19. 19 , 22 In the ICU, venous and arterial thromboembolic events may occur in up to 31% to 59% of patients with COVID-19. 16 , 22
Approximately 17% to 35% of hospitalized patients with COVID-19 are treated in an ICU, most commonly due to hypoxemic respiratory failure. Among patients in the ICU with COVID-19, 29% to 91% require invasive mechanical ventilation. 47 , 49 , 55 , 56 In addition to respiratory failure, hospitalized patients may develop acute kidney injury (9%), liver dysfunction (19%), bleeding and coagulation dysfunction (10%-25%), and septic shock (6%). 18 , 19 , 23 , 49 , 56
Approximately 2% to 5% of individuals with laboratory-confirmed COVID-19 are younger than 18 years, with a median age of 11 years. Children with COVID-19 have milder symptoms that are predominantly limited to the upper respiratory tract, and rarely require hospitalization. It is unclear why children are less susceptible to COVID-19. Potential explanations include that children have less robust immune responses (ie, no cytokine storm), partial immunity from other viral exposures, and lower rates of exposure to SARS-CoV-2. Although most pediatric cases are mild, a small percentage (<7%) of children admitted to the hospital for COVID-19 develop severe disease requiring mechanical ventilation. 57 A rare multisystem inflammatory syndrome similar to Kawasaki disease has recently been described in children in Europe and North America with SARS-CoV-2 infection. 58 , 59 This multisystem inflammatory syndrome in children is uncommon (2 in 100 000 persons aged <21 years). 60
Diagnosis of COVID-19 is typically made using polymerase chain reaction testing via nasal swab ( Box 2 ). However, because of false-negative test result rates of SARS-CoV-2 PCR testing of nasal swabs, clinical, laboratory, and imaging findings may also be used to make a presumptive diagnosis.
Reverse transcription polymerase chain reaction–based SARS-CoV-2 RNA detection from respiratory samples (eg, nasopharynx) is the standard for diagnosis. However, the sensitivity of testing varies with timing of testing relative to exposure. One modeling study estimated sensitivity at 33% 4 days after exposure, 62% on the day of symptom onset, and 80% 3 days after symptom onset. 61 - 63 Factors contributing to false-negative test results include the adequacy of the specimen collection technique, time from exposure, and specimen source. Lower respiratory samples, such as bronchoalveolar lavage fluid, are more sensitive than upper respiratory samples. Among 1070 specimens collected from 205 patients with COVID-19 in China, bronchoalveolar lavage fluid specimens had the highest positive rates of SARS-CoV-2 PCR testing results (93%), followed by sputum (72%), nasal swabs (63%), and pharyngeal swabs (32%). 61 SARS-CoV-2 can also be detected in feces, but not in urine. 61 Saliva may be an alternative specimen source that requires less personal protective equipment and fewer swabs, but requires further validation. 64
Several serological tests can also aid in the diagnosis and measurement of responses to novel vaccines. 62 , 65 , 66 However, the presence of antibodies may not confer immunity because not all antibodies produced in response to infection are neutralizing. Whether and how frequently second infections with SARS-CoV-2 occur remain unknown. Whether presence of antibody changes susceptibility to subsequent infection or how long antibody protection lasts are unknown. IgM antibodies are detectable within 5 days of infection, with higher IgM levels during weeks 2 to 3 of illness, while an IgG response is first seen approximately 14 days after symptom onset. 62 , 65 Higher antibody titers occur with more severe disease. 66 Available serological assays include point-of-care assays and high throughput enzyme immunoassays. However, test performance, accuracy, and validity are variable. 67
A systematic review of 19 studies of 2874 patients who were mostly from China (mean age, 52 years), of whom 88% were hospitalized, reported the typical range of laboratory abnormalities seen in COVID-19, including elevated serum C-reactive protein (increased in >60% of patients), lactate dehydrogenase (increased in approximately 50%-60%), alanine aminotransferase (elevated in approximately 25%), and aspartate aminotransferase (approximately 33%). 24 Approximately 75% of patients had low albumin. 24 The most common hematological abnormality is lymphopenia (absolute lymphocyte count <1.0 × 10 9 /L), which is present in up to 83% of hospitalized patients with COVID-19. 44 , 50 In conjunction with coagulopathy, modest prolongation of prothrombin times (prolonged in >5% of patients), mild thrombocytopenia (present in approximately 30% of patients) and elevated D-dimer values (present in 43%-60% of patients) are common. 14 , 15 , 19 , 44 , 68 However, most of these laboratory characteristics are nonspecific and are common in pneumonia. More severe laboratory abnormalities have been associated with more severe infection. 44 , 50 , 69 D-dimer and, to a lesser extent, lymphopenia seem to have the largest prognostic associations. 69
The characteristic chest computed tomographic imaging abnormalities for COVID-19 are diffuse, peripheral ground-glass opacities ( Figure 3 ). 70 Ground-glass opacities have ill-defined margins, air bronchograms, smooth or irregular interlobular or septal thickening, and thickening of the adjacent pleura. 70 Early in the disease, chest computed tomographic imaging findings in approximately 15% of individuals and chest radiograph findings in approximately 40% of individuals can be normal. 44 Rapid evolution of abnormalities can occur in the first 2 weeks after symptom onset, after which they subside gradually. 70 , 71
Chest computed tomographic imaging findings are nonspecific and overlap with other infections, so the diagnostic value of chest computed tomographic imaging for COVID-19 is limited. Some patients admitted to the hospital with polymerase chain reaction testing–confirmed SARS-CoV-2 infection have normal computed tomographic imaging findings, while abnormal chest computed tomographic imaging findings compatible with COVID-19 occur days before detection of SARS-CoV-2 RNA in other patients. 70 , 71
Currently, best practices for supportive management of acute hypoxic respiratory failure and ARDS should be followed. 72 - 74 Evidence-based guideline initiatives have been established by many countries and professional societies, 72 - 74 including guidelines that are updated regularly by the National Institutes of Health. 74
More than 75% of patients hospitalized with COVID-19 require supplemental oxygen therapy. For patients who are unresponsive to conventional oxygen therapy, heated high-flow nasal canula oxygen may be administered. 72 For patients requiring invasive mechanical ventilation, lung-protective ventilation with low tidal volumes (4-8 mL/kg, predicted body weight) and plateau pressure less than 30 mg Hg is recommended. 72 Additionally, prone positioning, a higher positive end-expiratory pressure strategy, and short-term neuromuscular blockade with cisatracurium or other muscle relaxants may facilitate oxygenation. Although some patients with COVID-19–related respiratory failure have high lung compliance, 75 they are still likely to benefit from lung-protective ventilation. 76 Cohorts of patients with ARDS have displayed similar heterogeneity in lung compliance, and even patients with greater compliance have shown benefit from lower tidal volume strategies. 76
The threshold for intubation in COVID-19–related respiratory failure is controversial, because many patients have normal work of breathing but severe hypoxemia. 77 “Earlier” intubation allows time for a more controlled intubation process, which is important given the logistical challenges of moving patients to an airborne isolation room and donning personal protective equipment prior to intubation. However, hypoxemia in the absence of respiratory distress is well tolerated, and patients may do well without mechanical ventilation. Earlier intubation thresholds may result in treating some patients with mechanical ventilation unnecessarily and exposing them to additional complications. Currently, insufficient evidence exists to make recommendations regarding earlier vs later intubation.
In observational studies, approximately 8% of hospitalized patients with COVID-19 experience a bacterial or fungal co-infection, but up to 72% are treated with broad-spectrum antibiotics. 78 Awaiting further data, it may be prudent to withhold antibacterial drugs in patients with COVID-19 and reserve them for those who present with radiological findings and/or inflammatory markers compatible with co-infection or who are immunocompromised and/or critically ill. 72
The following classes of drugs are being evaluated or developed for the management of COVID-19: antivirals (eg, remdesivir, favipiravir), antibodies (eg, convalescent plasma, hyperimmune immunoglobulins), anti-inflammatory agents (dexamethasone, statins), targeted immunomodulatory therapies (eg, tocilizumab, sarilumab, anakinra, ruxolitinib), anticoagulants (eg, heparin), and antifibrotics (eg, tyrosine kinase inhibitors). It is likely that different treatment modalities might have different efficacies at different stages of illness and in different manifestations of disease. Viral inhibition would be expected to be most effective early in infection, while, in hospitalized patients, immunomodulatory agents may be useful to prevent disease progression and anticoagulants may be useful to prevent thromboembolic complications.
More than 200 trials of chloroquine/hydroxychloroquine, compounds that inhibit viral entry and endocytosis of SARS-CoV-2 in vitro and may have beneficial immunomodulatory effects in vivo, 79 , 80 have been initiated, but early data from clinical trials in hospitalized patients with COVID-19 have not demonstrated clear benefit. 81 - 83 A clinical trial of 150 patients in China admitted to the hospital for mild to moderate COVID-19 did not find an effect on negative conversion of SARS-CoV-2 by 28 days (the main outcome measure) when compared with standard of care alone. 83 Two retrospective studies found no effect of hydroxychloroquine on risk of intubation or mortality among patients hospitalized for COVID-19. 84 , 85 One of these retrospective multicenter cohort studies compared in-hospital mortality between those treated with hydroxychloroquine plus azithromycin (735 patients), hydroxychloroquine alone (271 patients), azithromycin alone (211 patients), and neither drug (221 patients), but reported no differences across the groups. 84 Adverse effects are common, most notably QT prolongation with an increased risk of cardiac complications in an already vulnerable population. 82 , 84 These findings do not support off-label use of (hydroxy)chloroquine either with or without the coadministration of azithromycin. Randomized clinical trials are ongoing and should provide more guidance.
Most antiviral drugs undergoing clinical testing in patients with COVID-19 are repurposed antiviral agents originally developed against influenza, HIV, Ebola, or SARS/MERS. 79 , 86 Use of the protease inhibitor lopinavir-ritonavir, which disrupts viral replication in vitro, did not show benefit when compared with standard care in a randomized, controlled, open-label trial of 199 hospitalized adult patients with severe COVID-19. 87 Among the RNA-dependent RNA polymerase inhibitors, which halt SARS-CoV-2 replication, being evaluated, including ribavirin, favipiravir, and remdesivir, the latter seems to be the most promising. 79 , 88 The first preliminary results of a double-blind, randomized, placebo-controlled trial of 1063 adults hospitalized with COVID-19 and evidence of lower respiratory tract involvement who were randomly assigned to receive intravenous remdesivir or placebo for up to 10 days demonstrated that patients randomized to receive remdesivir had a shorter time to recovery than patients in the placebo group (11 vs 15 days). 88 A separate randomized, open-label trial among 397 hospitalized patients with COVID-19 who did not require mechanical ventilation reported that 5 days of treatment with remdesivir was not different than 10 days in terms of clinical status on day 14. 89 The effect of remdesivir on survival remains unknown.
Treatment with plasma obtained from patients who have recovered from viral infections was first reported during the 1918 flu pandemic. A first report of 5 critically ill patients with COVID-19 treated with convalescent plasma containing neutralizing antibody showed improvement in clinical status among all participants, defined as a combination of changes of body temperature, Sequential Organ Failure Assessment score, partial pressure of oxygen/fraction of inspired oxygen, viral load, serum antibody titer, routine blood biochemical index, ARDS, and ventilatory and extracorporeal membrane oxygenation supports before and after convalescent plasma transfusion status. 90 However, a subsequent multicenter, open-label, randomized clinical trial of 103 patients in China with severe COVID-19 found no statistical difference in time to clinical improvement within 28 days among patients randomized to receive convalescent plasma vs standard treatment alone (51.9% vs 43.1%). 91 However, the trial was stopped early because of slowing enrollment, which limited the power to detect a clinically important difference. Alternative approaches being studied include the use of convalescent plasma-derived hyperimmune globulin and monoclonal antibodies targeting SARS-CoV-2. 92 , 93
Alternative therapeutic strategies consist of modulating the inflammatory response in patients with COVID-19. Monoclonal antibodies directed against key inflammatory mediators, such as interferon gamma, interleukin 1, interleukin 6, and complement factor 5a, all target the overwhelming inflammatory response following SARS-CoV-2 infection with the goal of preventing organ damage. 79 , 86 , 94 Of these, the interleukin 6 inhibitors tocilizumab and sarilumab are best studied, with more than a dozen randomized clinical trials underway. 94 Tyrosine kinase inhibitors, such as imatinib, are studied for their potential to prevent pulmonary vascular leakage in individuals with COVID-19.
Studies of corticosteroids for viral pneumonia and ARDS have yielded mixed results. 72 , 73 However, the Randomized Evaluation of COVID-19 Therapy (RECOVERY) trial, which randomized 2104 patients with COVID-19 to receive 6 mg daily of dexamethasone for up to 10 days and 4321 to receive usual care, found that dexamethasone reduced 28-day all-cause mortality (21.6% vs 24.6%; age-adjusted rate ratio, 0.83 [95% CI, 0.74-0.92]; P < .001). 95 The benefit was greatest in patients with symptoms for more than 7 days and patients who required mechanical ventilation. By contrast, there was no benefit (and possibility for harm) among patients with shorter symptom duration and no supplemental oxygen requirement. A retrospective cohort study of 201 patients in Wuhan, China, with confirmed COVID-19 pneumonia and ARDS reported that treatment with methylprednisolone was associated with reduced risk of death (hazard ratio, 0.38 [95% CI, 0.20-0.72]). 69
Thromboembolic prophylaxis with subcutaneous low molecular weight heparin is recommended for all hospitalized patients with COVID-19. 15 , 19 Studies are ongoing to assess whether certain patients (ie, those with elevated D-dimer) benefit from therapeutic anticoagulation.
A disproportionate percentage of COVID-19 hospitalizations and deaths occurs in lower-income and minority populations. 45 , 96 , 97 In a report by the Centers for Disease Control and Prevention of 580 hospitalized patients for whom race data were available, 33% were Black and 45% were White, while 18% of residents in the surrounding community were Black and 59% were White. 45 The disproportionate prevalence of COVID-19 among Black patients was separately reported in a retrospective cohort study of 3626 patients with COVID-19 from Louisiana, in which 77% of patients hospitalized with COVID-19 and 71% of patients who died of COVID-19 were Black, but Black individuals comprised only 31% of the area population. 97 , 98 This disproportionate burden may be a reflection of disparities in housing, transportation, employment, and health. Minority populations are more likely to live in densely populated communities or housing, depend on public transportation, or work in jobs for which telework was not possible (eg, bus driver, food service worker). Black individuals also have a higher prevalence of chronic health conditions than White individuals. 98 , 99
Overall hospital mortality from COVID-19 is approximately 15% to 20%, but up to 40% among patients requiring ICU admission. However, mortality rates vary across cohorts, reflecting differences in the completeness of testing and case identification, variable thresholds for hospitalization, and differences in outcomes. Hospital mortality ranges from less than 5% among patients younger than 40 years to 35% for patients aged 70 to 79 years and greater than 60% for patients aged 80 to 89 years. 46 Estimated overall death rates by age group per 1000 confirmed cases are provided in the Table . Because not all people who die during the pandemic are tested for COVID-19, actual numbers of deaths from COVID-19 are higher than reported numbers.
Although long-term outcomes from COVID-19 are currently unknown, patients with severe illness are likely to suffer substantial sequelae. Survival from sepsis is associated with increased risk for mortality for at least 2 years, new physical disability, new cognitive impairment, and increased vulnerability to recurrent infection and further health deterioration. Similar sequalae are likely to be seen in survivors of severe COVID-19. 100
COVID-19 is a potentially preventable disease. The relationship between the intensity of public health action and the control of transmission is clear from the epidemiology of infection around the world. 25 , 101 , 102 However, because most countries have implemented multiple infection control measures, it is difficult to determine the relative benefit of each. 103 , 104 This question is increasingly important because continued interventions will be required until effective vaccines or treatments become available. In general, these interventions can be divided into those consisting of personal actions (eg, physical distancing, personal hygiene, and use of protective equipment), case and contact identification (eg, test-trace-track-isolate, reactive school or workplace closure), regulatory actions (eg, governmental limits on sizes of gatherings or business capacity; stay-at-home orders; proactive school, workplace, and public transport closure or restriction; cordon sanitaire or internal border closures), and international border measures (eg, border closure or enforced quarantine). A key priority is to identify the combination of measures that minimizes societal and economic disruption while adequately controlling infection. Optimal measures may vary between countries based on resource limitations, geography (eg, island nations and international border measures), population, and political factors (eg, health literacy, trust in government, cultural and linguistic diversity).
The evidence underlying these public health interventions has not changed since the 1918 flu pandemic. 105 Mathematical modeling studies and empirical evidence support that public health interventions, including home quarantine after infection, restricting mass gatherings, travel restrictions, and social distancing, are associated with reduced rates of transmission. 101 , 102 , 106 Risk of resurgence follows when these interventions are lifted.
A human vaccine is currently not available for SARS-CoV-2, but approximately 120 candidates are under development. Approaches include the use of nucleic acids (DNA or RNA), inactivated or live attenuated virus, viral vectors, and recombinant proteins or virus particles. 107 , 108 Challenges to developing an effective vaccine consist of technical barriers (eg, whether S or receptor-binding domain proteins provoke more protective antibodies, prior exposure to adenovirus serotype 5 [which impairs immunogenicity in the viral vector vaccine], need for adjuvant), feasibility of large-scale production and regulation (eg, ensuring safety and effectiveness), and legal barriers (eg, technology transfer and licensure agreements). The SARS-CoV-2 S protein appears to be a promising immunogen for protection, but whether targeting the full-length protein or only the receptor-binding domain is sufficient to prevent transmission remains unclear. 108 Other considerations include the potential duration of immunity and thus the number of vaccine doses needed to confer immunity. 62 , 108 More than a dozen candidate SARS-CoV-2 vaccines are currently being tested in phase 1-3 trials.
Other approaches to prevention are likely to emerge in the coming months, including monoclonal antibodies, hyperimmune globulin, and convalscent titer. If proved effective, these approaches could be used in high-risk individuals, including health care workers, other essential workers, and older adults (particularly those in nursing homes or long-term care facilities).
This review has several limitations. First, information regarding SARS CoV-2 is limited. Second, information provided here is based on current evidence, but may be modified as more information becomes available. Third, few randomized trials have been published to guide management of COVID-19.
As of July 1, 2020, more than 10 million people worldwide had been infected with SARS-CoV-2. Many aspects of transmission, infection, and treatment remain unclear. Advances in prevention and effective management of COVID-19 will require basic and clinical investigation and public health and clinical interventions.
Accepted for Publication: June 30, 2020.
Corresponding Author: W. Joost Wiersinga, MD, PhD, Division of Infectious Diseases, Department of Medicine, Amsterdam UMC, location AMC, Meibergdreef 9, 1105 AZ Amsterdam, the Netherlands ( [email protected] ).
Published Online: July 10, 2020. doi:10.1001/jama.2020.12839
Conflict of Interest Disclosures: Dr Wiersinga is supported by the Netherlands Organisation of Scientific Research outside the submitted work. Dr Prescott reported receiving grants from the US Agency for Healthcare Research and Quality (HCP by R01 HS026725), the National Institutes of Health/National Institute of General Medical Sciences, and the US Department of Veterans Affairs outside the submitted work, being the sepsis physician lead for the Hospital Medicine Safety Continuous Quality Initiative funded by BlueCross/BlueShield of Michigan, and serving on the steering committee for MI-COVID-19, a Michigan statewide registry to improve care for patients with COVID-19 in Michigan. Dr Rhodes reported being the co-chair of the Surviving Sepsis Campaign. Dr Cheng reported being a member of Australian government advisory committees, including those involved in COVID-19. No other disclosures were reported.
Disclaimer: This article does not represent the views of the US Department of Veterans Affairs or the US government. This material is the result of work supported with resources and use of facilities at the Ann Arbor VA Medical Center. The opinions in this article do not necessarily represent those of the Australian government or advisory committees.
- Register for email alerts with links to free full-text articles
- Access PDFs of free articles
- Manage your interests
- Save searches and receive search alerts
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- Springer Nature - PMC COVID-19 Collection

A Review of Coronavirus Disease-2019 (COVID-19)
Tanu singhal.
Department of Pediatrics and Infectious Disease, Kokilaben Dhirubhai Ambani Hospital and Medical Research Institute, Mumbai, India
There is a new public health crises threatening the world with the emergence and spread of 2019 novel coronavirus (2019-nCoV) or the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The virus originated in bats and was transmitted to humans through yet unknown intermediary animals in Wuhan, Hubei province, China in December 2019. There have been around 96,000 reported cases of coronavirus disease 2019 (COVID-2019) and 3300 reported deaths to date (05/03/2020). The disease is transmitted by inhalation or contact with infected droplets and the incubation period ranges from 2 to 14 d. The symptoms are usually fever, cough, sore throat, breathlessness, fatigue, malaise among others. The disease is mild in most people; in some (usually the elderly and those with comorbidities), it may progress to pneumonia, acute respiratory distress syndrome (ARDS) and multi organ dysfunction. Many people are asymptomatic. The case fatality rate is estimated to range from 2 to 3%. Diagnosis is by demonstration of the virus in respiratory secretions by special molecular tests. Common laboratory findings include normal/ low white cell counts with elevated C-reactive protein (CRP). The computerized tomographic chest scan is usually abnormal even in those with no symptoms or mild disease. Treatment is essentially supportive; role of antiviral agents is yet to be established. Prevention entails home isolation of suspected cases and those with mild illnesses and strict infection control measures at hospitals that include contact and droplet precautions. The virus spreads faster than its two ancestors the SARS-CoV and Middle East respiratory syndrome coronavirus (MERS-CoV), but has lower fatality. The global impact of this new epidemic is yet uncertain.
Introduction
The 2019 novel coronavirus (2019-nCoV) or the severe acute respiratory syndrome corona virus 2 (SARS-CoV-2) as it is now called, is rapidly spreading from its origin in Wuhan City of Hubei Province of China to the rest of the world [ 1 ]. Till 05/03/2020 around 96,000 cases of coronavirus disease 2019 (COVID-19) and 3300 deaths have been reported [ 2 ]. India has reported 29 cases till date. Fortunately so far, children have been infrequently affected with no deaths. But the future course of this virus is unknown. This article gives a bird’s eye view about this new virus. Since knowledge about this virus is rapidly evolving, readers are urged to update themselves regularly.
Coronaviruses are enveloped positive sense RNA viruses ranging from 60 nm to 140 nm in diameter with spike like projections on its surface giving it a crown like appearance under the electron microscope; hence the name coronavirus [ 3 ]. Four corona viruses namely HKU1, NL63, 229E and OC43 have been in circulation in humans, and generally cause mild respiratory disease.
There have been two events in the past two decades wherein crossover of animal betacorona viruses to humans has resulted in severe disease. The first such instance was in 2002–2003 when a new coronavirus of the β genera and with origin in bats crossed over to humans via the intermediary host of palm civet cats in the Guangdong province of China. This virus, designated as severe acute respiratory syndrome coronavirus affected 8422 people mostly in China and Hong Kong and caused 916 deaths (mortality rate 11%) before being contained [ 4 ]. Almost a decade later in 2012, the Middle East respiratory syndrome coronavirus (MERS-CoV), also of bat origin, emerged in Saudi Arabia with dromedary camels as the intermediate host and affected 2494 people and caused 858 deaths (fatality rate 34%) [ 5 ].
Origin and Spread of COVID-19 [ 1 , 2 , 6 ]
In December 2019, adults in Wuhan, capital city of Hubei province and a major transportation hub of China started presenting to local hospitals with severe pneumonia of unknown cause. Many of the initial cases had a common exposure to the Huanan wholesale seafood market that also traded live animals. The surveillance system (put into place after the SARS outbreak) was activated and respiratory samples of patients were sent to reference labs for etiologic investigations. On December 31st 2019, China notified the outbreak to the World Health Organization and on 1st January the Huanan sea food market was closed. On 7th January the virus was identified as a coronavirus that had >95% homology with the bat coronavirus and > 70% similarity with the SARS- CoV. Environmental samples from the Huanan sea food market also tested positive, signifying that the virus originated from there [ 7 ]. The number of cases started increasing exponentially, some of which did not have exposure to the live animal market, suggestive of the fact that human-to-human transmission was occurring [ 8 ]. The first fatal case was reported on 11th Jan 2020. The massive migration of Chinese during the Chinese New Year fuelled the epidemic. Cases in other provinces of China, other countries (Thailand, Japan and South Korea in quick succession) were reported in people who were returning from Wuhan. Transmission to healthcare workers caring for patients was described on 20th Jan, 2020. By 23rd January, the 11 million population of Wuhan was placed under lock down with restrictions of entry and exit from the region. Soon this lock down was extended to other cities of Hubei province. Cases of COVID-19 in countries outside China were reported in those with no history of travel to China suggesting that local human-to-human transmission was occurring in these countries [ 9 ]. Airports in different countries including India put in screening mechanisms to detect symptomatic people returning from China and placed them in isolation and testing them for COVID-19. Soon it was apparent that the infection could be transmitted from asymptomatic people and also before onset of symptoms. Therefore, countries including India who evacuated their citizens from Wuhan through special flights or had travellers returning from China, placed all people symptomatic or otherwise in isolation for 14 d and tested them for the virus.
Cases continued to increase exponentially and modelling studies reported an epidemic doubling time of 1.8 d [ 10 ]. In fact on the 12th of February, China changed its definition of confirmed cases to include patients with negative/ pending molecular tests but with clinical, radiologic and epidemiologic features of COVID-19 leading to an increase in cases by 15,000 in a single day [ 6 ]. As of 05/03/2020 96,000 cases worldwide (80,000 in China) and 87 other countries and 1 international conveyance (696, in the cruise ship Diamond Princess parked off the coast of Japan) have been reported [ 2 ]. It is important to note that while the number of new cases has reduced in China lately, they have increased exponentially in other countries including South Korea, Italy and Iran. Of those infected, 20% are in critical condition, 25% have recovered, and 3310 (3013 in China and 297 in other countries) have died [ 2 ]. India, which had reported only 3 cases till 2/3/2020, has also seen a sudden spurt in cases. By 5/3/2020, 29 cases had been reported; mostly in Delhi, Jaipur and Agra in Italian tourists and their contacts. One case was reported in an Indian who traveled back from Vienna and exposed a large number of school children in a birthday party at a city hotel. Many of the contacts of these cases have been quarantined.
These numbers are possibly an underestimate of the infected and dead due to limitations of surveillance and testing. Though the SARS-CoV-2 originated from bats, the intermediary animal through which it crossed over to humans is uncertain. Pangolins and snakes are the current suspects.
Epidemiology and Pathogenesis [ 10 , 11 ]
All ages are susceptible. Infection is transmitted through large droplets generated during coughing and sneezing by symptomatic patients but can also occur from asymptomatic people and before onset of symptoms [ 9 ]. Studies have shown higher viral loads in the nasal cavity as compared to the throat with no difference in viral burden between symptomatic and asymptomatic people [ 12 ]. Patients can be infectious for as long as the symptoms last and even on clinical recovery. Some people may act as super spreaders; a UK citizen who attended a conference in Singapore infected 11 other people while staying in a resort in the French Alps and upon return to the UK [ 6 ]. These infected droplets can spread 1–2 m and deposit on surfaces. The virus can remain viable on surfaces for days in favourable atmospheric conditions but are destroyed in less than a minute by common disinfectants like sodium hypochlorite, hydrogen peroxide etc. [ 13 ]. Infection is acquired either by inhalation of these droplets or touching surfaces contaminated by them and then touching the nose, mouth and eyes. The virus is also present in the stool and contamination of the water supply and subsequent transmission via aerosolization/feco oral route is also hypothesized [ 6 ]. As per current information, transplacental transmission from pregnant women to their fetus has not been described [ 14 ]. However, neonatal disease due to post natal transmission is described [ 14 ]. The incubation period varies from 2 to 14 d [median 5 d]. Studies have identified angiotensin receptor 2 (ACE 2 ) as the receptor through which the virus enters the respiratory mucosa [ 11 ].
The basic case reproduction rate (BCR) is estimated to range from 2 to 6.47 in various modelling studies [ 11 ]. In comparison, the BCR of SARS was 2 and 1.3 for pandemic flu H1N1 2009 [ 2 ].
Clinical Features [ 8 , 15 – 18 ]
The clinical features of COVID-19 are varied, ranging from asymptomatic state to acute respiratory distress syndrome and multi organ dysfunction. The common clinical features include fever (not in all), cough, sore throat, headache, fatigue, headache, myalgia and breathlessness. Conjunctivitis has also been described. Thus, they are indistinguishable from other respiratory infections. In a subset of patients, by the end of the first week the disease can progress to pneumonia, respiratory failure and death. This progression is associated with extreme rise in inflammatory cytokines including IL2, IL7, IL10, GCSF, IP10, MCP1, MIP1A, and TNFα [ 15 ]. The median time from onset of symptoms to dyspnea was 5 d, hospitalization 7 d and acute respiratory distress syndrome (ARDS) 8 d. The need for intensive care admission was in 25–30% of affected patients in published series. Complications witnessed included acute lung injury, ARDS, shock and acute kidney injury. Recovery started in the 2nd or 3rd wk. The median duration of hospital stay in those who recovered was 10 d. Adverse outcomes and death are more common in the elderly and those with underlying co-morbidities (50–75% of fatal cases). Fatality rate in hospitalized adult patients ranged from 4 to 11%. The overall case fatality rate is estimated to range between 2 and 3% [ 2 ].
Interestingly, disease in patients outside Hubei province has been reported to be milder than those from Wuhan [ 17 ]. Similarly, the severity and case fatality rate in patients outside China has been reported to be milder [ 6 ]. This may either be due to selection bias wherein the cases reporting from Wuhan included only the severe cases or due to predisposition of the Asian population to the virus due to higher expression of ACE 2 receptors on the respiratory mucosa [ 11 ].
Disease in neonates, infants and children has been also reported to be significantly milder than their adult counterparts. In a series of 34 children admitted to a hospital in Shenzhen, China between January 19th and February 7th, there were 14 males and 20 females. The median age was 8 y 11 mo and in 28 children the infection was linked to a family member and 26 children had history of travel/residence to Hubei province in China. All the patients were either asymptomatic (9%) or had mild disease. No severe or critical cases were seen. The most common symptoms were fever (50%) and cough (38%). All patients recovered with symptomatic therapy and there were no deaths. One case of severe pneumonia and multiorgan dysfunction in a child has also been reported [ 19 ]. Similarly the neonatal cases that have been reported have been mild [ 20 ].
Diagnosis [ 21 ]
A suspect case is defined as one with fever, sore throat and cough who has history of travel to China or other areas of persistent local transmission or contact with patients with similar travel history or those with confirmed COVID-19 infection. However cases may be asymptomatic or even without fever. A confirmed case is a suspect case with a positive molecular test.
Specific diagnosis is by specific molecular tests on respiratory samples (throat swab/ nasopharyngeal swab/ sputum/ endotracheal aspirates and bronchoalveolar lavage). Virus may also be detected in the stool and in severe cases, the blood. It must be remembered that the multiplex PCR panels currently available do not include the COVID-19. Commercial tests are also not available at present. In a suspect case in India, the appropriate sample has to be sent to designated reference labs in India or the National Institute of Virology in Pune. As the epidemic progresses, commercial tests will become available.
Other laboratory investigations are usually non specific. The white cell count is usually normal or low. There may be lymphopenia; a lymphocyte count <1000 has been associated with severe disease. The platelet count is usually normal or mildly low. The CRP and ESR are generally elevated but procalcitonin levels are usually normal. A high procalcitonin level may indicate a bacterial co-infection. The ALT/AST, prothrombin time, creatinine, D-dimer, CPK and LDH may be elevated and high levels are associated with severe disease.
The chest X-ray (CXR) usually shows bilateral infiltrates but may be normal in early disease. The CT is more sensitive and specific. CT imaging generally shows infiltrates, ground glass opacities and sub segmental consolidation. It is also abnormal in asymptomatic patients/ patients with no clinical evidence of lower respiratory tract involvement. In fact, abnormal CT scans have been used to diagnose COVID-19 in suspect cases with negative molecular diagnosis; many of these patients had positive molecular tests on repeat testing [ 22 ].
Differential Diagnosis [ 21 ]
The differential diagnosis includes all types of respiratory viral infections [influenza, parainfluenza, respiratory syncytial virus (RSV), adenovirus, human metapneumovirus, non COVID-19 coronavirus], atypical organisms (mycoplasma, chlamydia) and bacterial infections. It is not possible to differentiate COVID-19 from these infections clinically or through routine lab tests. Therefore travel history becomes important. However, as the epidemic spreads, the travel history will become irrelevant.
Treatment [ 21 , 23 ]
Treatment is essentially supportive and symptomatic.
The first step is to ensure adequate isolation (discussed later) to prevent transmission to other contacts, patients and healthcare workers. Mild illness should be managed at home with counseling about danger signs. The usual principles are maintaining hydration and nutrition and controlling fever and cough. Routine use of antibiotics and antivirals such as oseltamivir should be avoided in confirmed cases. In hypoxic patients, provision of oxygen through nasal prongs, face mask, high flow nasal cannula (HFNC) or non-invasive ventilation is indicated. Mechanical ventilation and even extra corporeal membrane oxygen support may be needed. Renal replacement therapy may be needed in some. Antibiotics and antifungals are required if co-infections are suspected or proven. The role of corticosteroids is unproven; while current international consensus and WHO advocate against their use, Chinese guidelines do recommend short term therapy with low-to-moderate dose corticosteroids in COVID-19 ARDS [ 24 , 25 ]. Detailed guidelines for critical care management for COVID-19 have been published by the WHO [ 26 ]. There is, as of now, no approved treatment for COVID-19. Antiviral drugs such as ribavirin, lopinavir-ritonavir have been used based on the experience with SARS and MERS. In a historical control study in patients with SARS, patients treated with lopinavir-ritonavir with ribavirin had better outcomes as compared to those given ribavirin alone [ 15 ].
In the case series of 99 hospitalized patients with COVID-19 infection from Wuhan, oxygen was given to 76%, non-invasive ventilation in 13%, mechanical ventilation in 4%, extracorporeal membrane oxygenation (ECMO) in 3%, continuous renal replacement therapy (CRRT) in 9%, antibiotics in 71%, antifungals in 15%, glucocorticoids in 19% and intravenous immunoglobulin therapy in 27% [ 15 ]. Antiviral therapy consisting of oseltamivir, ganciclovir and lopinavir-ritonavir was given to 75% of the patients. The duration of non-invasive ventilation was 4–22 d [median 9 d] and mechanical ventilation for 3–20 d [median 17 d]. In the case series of children discussed earlier, all children recovered with basic treatment and did not need intensive care [ 17 ].
There is anecdotal experience with use of remdeswir, a broad spectrum anti RNA drug developed for Ebola in management of COVID-19 [ 27 ]. More evidence is needed before these drugs are recommended. Other drugs proposed for therapy are arbidol (an antiviral drug available in Russia and China), intravenous immunoglobulin, interferons, chloroquine and plasma of patients recovered from COVID-19 [ 21 , 28 , 29 ]. Additionally, recommendations about using traditional Chinese herbs find place in the Chinese guidelines [ 21 ].
Prevention [ 21 , 30 ]
Since at this time there are no approved treatments for this infection, prevention is crucial. Several properties of this virus make prevention difficult namely, non-specific features of the disease, the infectivity even before onset of symptoms in the incubation period, transmission from asymptomatic people, long incubation period, tropism for mucosal surfaces such as the conjunctiva, prolonged duration of the illness and transmission even after clinical recovery.
Isolation of confirmed or suspected cases with mild illness at home is recommended. The ventilation at home should be good with sunlight to allow for destruction of virus. Patients should be asked to wear a simple surgical mask and practice cough hygiene. Caregivers should be asked to wear a surgical mask when in the same room as patient and use hand hygiene every 15–20 min.
The greatest risk in COVID-19 is transmission to healthcare workers. In the SARS outbreak of 2002, 21% of those affected were healthcare workers [ 31 ]. Till date, almost 1500 healthcare workers in China have been infected with 6 deaths. The doctor who first warned about the virus has died too. It is important to protect healthcare workers to ensure continuity of care and to prevent transmission of infection to other patients. While COVID-19 transmits as a droplet pathogen and is placed in Category B of infectious agents (highly pathogenic H5N1 and SARS), by the China National Health Commission, infection control measures recommended are those for category A agents (cholera, plague). Patients should be placed in separate rooms or cohorted together. Negative pressure rooms are not generally needed. The rooms and surfaces and equipment should undergo regular decontamination preferably with sodium hypochlorite. Healthcare workers should be provided with fit tested N95 respirators and protective suits and goggles. Airborne transmission precautions should be taken during aerosol generating procedures such as intubation, suction and tracheostomies. All contacts including healthcare workers should be monitored for development of symptoms of COVID-19. Patients can be discharged from isolation once they are afebrile for atleast 3 d and have two consecutive negative molecular tests at 1 d sampling interval. This recommendation is different from pandemic flu where patients were asked to resume work/school once afebrile for 24 h or by day 7 of illness. Negative molecular tests were not a prerequisite for discharge.
At the community level, people should be asked to avoid crowded areas and postpone non-essential travel to places with ongoing transmission. They should be asked to practice cough hygiene by coughing in sleeve/ tissue rather than hands and practice hand hygiene frequently every 15–20 min. Patients with respiratory symptoms should be asked to use surgical masks. The use of mask by healthy people in public places has not shown to protect against respiratory viral infections and is currently not recommended by WHO. However, in China, the public has been asked to wear masks in public and especially in crowded places and large scale gatherings are prohibited (entertainment parks etc). China is also considering introducing legislation to prohibit selling and trading of wild animals [ 32 ].
The international response has been dramatic. Initially, there were massive travel restrictions to China and people returning from China/ evacuated from China are being evaluated for clinical symptoms, isolated and tested for COVID-19 for 2 wks even if asymptomatic. However, now with rapid world wide spread of the virus these travel restrictions have extended to other countries. Whether these efforts will lead to slowing of viral spread is not known.
A candidate vaccine is under development.
Practice Points from an Indian Perspective
At the time of writing this article, the risk of coronavirus in India is extremely low. But that may change in the next few weeks. Hence the following is recommended:
- Healthcare providers should take travel history of all patients with respiratory symptoms, and any international travel in the past 2 wks as well as contact with sick people who have travelled internationally.
- They should set up a system of triage of patients with respiratory illness in the outpatient department and give them a simple surgical mask to wear. They should use surgical masks themselves while examining such patients and practice hand hygiene frequently.
- Suspected cases should be referred to government designated centres for isolation and testing (in Mumbai, at this time, it is Kasturba hospital). Commercial kits for testing are not yet available in India.
- Patients admitted with severe pneumonia and acute respiratory distress syndrome should be evaluated for travel history and placed under contact and droplet isolation. Regular decontamination of surfaces should be done. They should be tested for etiology using multiplex PCR panels if logistics permit and if no pathogen is identified, refer the samples for testing for SARS-CoV-2.
- All clinicians should keep themselves updated about recent developments including global spread of the disease.
- Non-essential international travel should be avoided at this time.
- People should stop spreading myths and false information about the disease and try to allay panic and anxiety of the public.
Conclusions
This new virus outbreak has challenged the economic, medical and public health infrastructure of China and to some extent, of other countries especially, its neighbours. Time alone will tell how the virus will impact our lives here in India. More so, future outbreaks of viruses and pathogens of zoonotic origin are likely to continue. Therefore, apart from curbing this outbreak, efforts should be made to devise comprehensive measures to prevent future outbreaks of zoonotic origin.
Compliance with Ethical Standards
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.

IMAGES
VIDEO
COMMENTS
Abstract. In early December 2019, an outbreak of coronavirus disease 2019 (COVID-19), caused by a novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), occurred in Wuhan City, Hubei Province, China. On January 30, 2020 the World Health Organization declared the outbreak as a Public Health Emergency of International Concern.
Coronavirus disease 2019 (COVID-19): A literature review.pdf. Content uploaded by Naoya Itoh. Author content. All content in this area was uploaded by Naoya Itoh on May 04, 2020 .
A systematic review was conducted to identify clinical trials of pharmacological interventions for COVID-19 published between 1 December 2019 and 14 October 2020. Data regarding efficacy of interventions, in terms of mortality, hospitalisation and need for ventilation, were extracted from identified studies and synthesised qualitatively.
Pediatric Research - A literature review of 2019 novel coronavirus (SARS-CoV2) infection in neonates and children. ... Download PDF. Download PDF. Review Article; Published: 17 July 2020;
reduced COVID-19 mortality by 0.2% on average. SIPOs were also ineffective, only reducing COVID-19 mortality by 2.9% on average. Specific NPI studies also find no broad-based evidence of noticeable effects on COVID-19 mortality. While this meta-analysis concludes that lockdowns have had little to no public health effects,
The global epidemiology of coronavirus disease 2019 (COVID-19) suggests a wide spectrum of clinical severity, ranging from asymptomatic to fatal. Al-though the clinical and laboratory characteristics of COVID-19 patients have been well characterized, the pathophysiological mechanisms underlying dis-ease severity and progression remain unclear.
Introduction. The coronavirus disease 2019 (COVID-19) pandemic caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has resulted in over 192 million cases and 4.1 million deaths as of July 22, 2021. 1 This pandemic has brought along a massive burden in morbidity and mortality in the healthcare systems. Despite the implementation of stringent public health measures, there ...
COVID-19, the disease caused by SARS-CoV-2, has claimed approximately 5 million lives and 257 million cases reported globally. This virus and disease have significantly affected people worldwide ...
We aimed to summarize and critically appraise systematic reviews of coronavirus disease (COVID-19) in humans that were available at the beginning of the pandemic. Global Research, LILACS, and Epistemonikos) were searched from December 1, 2019, to March 24, 2020. Systematic reviews analyzing primary studies of COVID-19 were included.
This literature review was conducted with an extensive search of databases, including PubMed, Web of Science (WOS), and Scopus, using the keywords "COVID19", "2019-nCoV disease", "2019 novel coronavirus infection", "Nurse", "NursingCare", and" Nursing management.". The span of the literature search was between December ...
Navigating the rapidly growing body of scientific literature on the SARS-CoV-2 pandemic is challenging, and ongoing critical appraisal of this output is essential. We aimed to summarize and critically appraise systematic reviews of coronavirus disease (COVID-19) in humans that were available at the beginning of the pandemic. Nine databases (Medline, EMBASE, Cochrane Library, CINAHL, Web of ...
Transmission. The role of the Huanan Seafood Wholesale Market in propagating disease is unclear. Many initial COVID-19 cases were linked to this market suggesting that SARS-CoV-2 was transmitted from animals to humans .However, a genomic study has provided evidence that the virus was introduced from another, yet unknown location, into the market where it spread more rapidly, although human-to ...
Specifically, we undertook a systematic review to identify COVID-19 clinical literature and matched them to historical controls to formally evaluate the following: (1) the methodological quality ...
This literature review highlights the dynamic nature of COVID-19 transmission and presentation. Analyzing 59 relevant articles up to May 1st, 2020 reflects that the main reported clinical manifestation of COVID-19 pandemic is fever and respiratory involvement. Also, current literature demonstrates a wide spectrum of different and atypical ...
Introduction: As the COVID-19 pandemic ravages the world, nursing resources, and capacities play an essential role in disease management. This literature review focuses on the central issues related to the nursing care of patients affected by COVID-19. Material and methods: This literature review was conducted with an extensive search of databases, including PubMed, Web of Science (WOS), and ...
Another systematic review of 30 studies involving 3834 COVID-19 patients revealed that overall co-infection rate in hospitalized patients was 7%. In a mixed setting of hospital ward and intensive care unit (ICU), the most common germs were Mycoplasma pneumoniae, Pseudomonas aeruginosa, and Haemophilus influenzae.
Abstract. In early December 2019, an outbreak of coronavirus disease 2019 (COVID-19), caused by a novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), occurred in Wuhan City, Hubei Province, China. On January 30, 2020 the World Health Organization declared the outbreak as a Public Health Emergency of International Concern.
(Additional file 1). We conducted a scoping review to summarize and critically analyze all the published scien-tific articles regarding the new coronavirus in January 2020. This review aims to provide the evidence of early findings on the epidemiology, causes, clinical diagnosis, as well as prevention and control of COVID-19 in relation to
Chu DK, Akl EA, Duda S, et al; COVID-19 Systematic Urgent Review Group Effort (SURGE) study authors. Physical distancing, face masks, and eye protection to prevent person-to-person transmission of SARS-CoV-2 and COVID-19: a systematic review and meta-analysis.
Keywords: children, COVID-19, coronavirus, physical activity, psychology. Citation: Cachón-Zagalaz J, Sánchez-Zafra M, Sanabrias-Moreno D, González-Valero G, Lara-Sánchez AJ and Zagalaz-Sánchez ML (2020) Systematic Review of the Literature About the Effects of the COVID-19 Pandemic on the Lives of School Children. Front.
literature review examined the question: What is the evidence for a relationship between human-animal interaction and/or animal ownership and common mental disorders among adults who interacted with pets compared to adults who did not during the COVID-19 pandemic? Methods: To address this question, four databases were searched: Medline,
Abstract. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is the seventh-generation coronavirus family causing viral pandemic coronavirus disease (COVID-19) across globe affecting millions of people. The objectives of this study are to (1) identify the major research themes in COVID-19 literature, (2) determine the origin, symptoms ...
COVID -19 Pandemic- A Literature Review. G. Singla, Tanisha Singla, S. Singla. Published 2020. Medicine. International Journal of Research. TLDR. To understand about corona virus disease, an extensive research of literature was done and the findings were summed up in this article. Expand. ijrrjournal.com.
Results indicate that the mental health of informal carers, already poorer pre-COVID compared to non-caregivers, was disproportionally impacted as a result of the pandemic and its associated public health containment measures. The COVID-19 pandemic imposed additional and specific challenges on the lives and wellbeing of informal unpaid carers. Addressing an important gap in the existing ...
There have been around 96,000 reported cases of coronavirus disease 2019 (COVID-2019) and 3300 reported deaths to date (05/03/2020). The disease is transmitted by inhalation or contact with infected droplets and the incubation period ranges from 2 to 14 d. The symptoms are usually fever, cough, sore throat, breathlessness, fatigue, malaise ...